Editor: Tiffany
A recent study demonstrates that complexing the natural hepatoprotector icariin with hydroxypropyl-γ-cyclodextrin significantly enhances its solubility and bioavailability, indicating improved therapeutic potential for liver health.
Key Highlights
- Research Question:
Can the solubility and pharmacokinetic profile of icariin, a natural hepatoprotector, be improved through complexation with HP-γ-cyclodextrin? - Research Difficulties:
Icariin’s clinical utility has been hampered by its extremely low water solubility (0.02 mg/mL) and challenges in achieving effective drug delivery. - Key Findings:
The study found that complexation increased icariin’s solubility by 654 times and enhanced its bioavailability nearly 20-fold, demonstrating a significant improvement in therapeutic potential. - Innovative Aspects:
The use of HP-γ-cyclodextrin and a trace amount of water-soluble polymer to achieve such a drastic increase in solubility and pharmacokinetics represents a novel approach in drug formulation. - Importance of the Study:
This research provides a potential pathway to develop more effective therapies for liver conditions and enhances the clinical applicability of icariin as a hepatoprotector and anti-liver cancer agent.
Icariin: A Natural Hepatoprotector with Limited Solubility
Icariin, an isoamyl flavonol glycoside extracted from the traditional Chinese medicinal herb Herba epimedii, has garnered significant attention in recent years due to its multiple pharmacological properties. Research has demonstrated that icariin possesses the ability to expand both cardiovascular and cerebral blood vessels, enhance hematopoietic functions, and strengthen the immune system. Furthermore, icariin is noted for its anti-aging effects and potential anti-cancer activities, particularly against liver tumors. Despite these promising benefits, its clinical application is severely limited by its extremely low solubility in water (0.02 mg/mL). This low solubility poses a significant barrier to its effective delivery and therapeutic efficacy. Various methods, including the use of cyclodextrins, have been explored to improve icariin’s solubility, but the results have been modest. There is an urgent need for innovative approaches that can enhance the bioavailability and therapeutic potential of icariin, paving the way for its use in clinical settings as a hepatoprotector and an adjunct in liver cancer therapy.
Objectives: Enhancing the Solubility and Pharmacokinetics of Icariin
This study aims to investigate the potential of complexing icariin with hydroxypropyl-γ-cyclodextrin (HP-γ-cyclodextrin) to significantly enhance its solubility and pharmacokinetic profile. The specific objectives of the research include:
- To evaluate the solubility of icariin in the presence of HP-γ-cyclodextrin: This objective involves systematically examining the phase solubility of icariin when complexed with various concentrations of HP-γ-cyclodextrin, utilizing methods such as the Higuchi-Connors equation to classify the interaction type.
- To optimize the complexation process: The research will explore different preparation methods—including aqueous solution, sonication, microwave, autoclave, and supercritical CO2—to identify the most effective technique for enhancing icariin’s solubility.
- To assess the pharmacokinetic parameters of the icariin-HP-γ-cyclodextrin complex: This includes evaluating the in vitro dissolution rates and conducting in vivo studies to measure key pharmacokinetic parameters such as Cmax, AUC, clearance, and half-life, thereby determining the efficacy and potential clinical applications of the complex as a hepatoprotector.
By achieving these objectives, the study aims to provide a comprehensive understanding of how complexation can enhance the therapeutic potential of icariin, ultimately contributing to the development of more effective treatments for liver-related health issues.
Methodological Approach: Experimental Design and Key Findings
I. Experimental Process
- Preparation of icariin and HP-γ-cyclodextrin solutions.
- Conduct phase solubility analysis with various cyclodextrin derivatives.
- Select HP-γ-cyclodextrin based on solubility results.
- Optimize complexation conditions using the solution method.
- Employ different methods (sonication, microwave, autoclave, and supercritical CO2) for complex preparation.
- Characterize the inclusion complex using FTIR, SEM, TGA, DSC, and XRD.
- Conduct in vitro dissolution tests of icariin, the physical mixture, and the inclusion complex.
- Perform in vivo pharmacokinetic studies in dogs with both icariin and the complex.
- Analyze plasma samples using HPLC to determine pharmacokinetic parameters.
II. Key Experiments
1. Phase Solubility Analysis
Procedure: Solutions of various cyclodextrins (β-cyclodextrin, HP-β-cyclodextrin, methyl-β-cyclodextrin, γ-cyclodextrin, and HP-γ-cyclodextrin) were prepared in different concentrations. Icariin was added to each solution and stirred for 48 hours at room temperature. Filtrates were analyzed by HPLC to determine the solubility of icariin.
Result: HP-γ-cyclodextrin exhibited the highest solubilization effect on icariin, achieving a significant increase in solubility compared to other cyclodextrins.
Finding: The phase solubility curve indicated that the system exhibited an “AL” type behavior, confirming the effective inclusion of icariin by HP-γ-cyclodextrin.
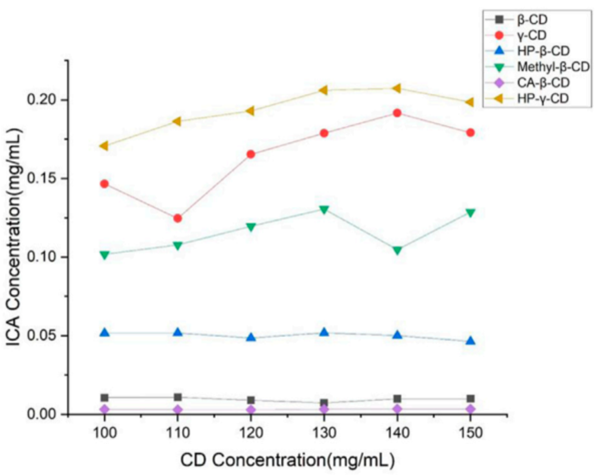
Figure 1. Phase solubility curves of icariin with β-cyclodextrin, HP-β-cyclodextrin, methyl-β-cyclodextrin, citric acid-β-cyclodextrin, γ-cyclodextrin and HP-γ-cyclodextrin.
2. Optimization of Complexation Conditions
Procedure: The solution method was utilized to optimize the molar ratio of icariin to HP-γ-cyclodextrin and the stirring temperature. Various ratios (1:1, 1:2, and 1:3) and temperatures (40°C, 50°C, and 60°C) were tested. The solution was stirred for varying durations (6 to 12 hours).
Result: The optimal conditions yielded a maximum solubility of icariin at a 1:1 molar ratio and a stirring temperature of 50°C for 10 hours.
Finding: Under these conditions, icariin’s solubility increased to approximately 7.83 mg/mL, marking a significant enhancement over the initial solubility.
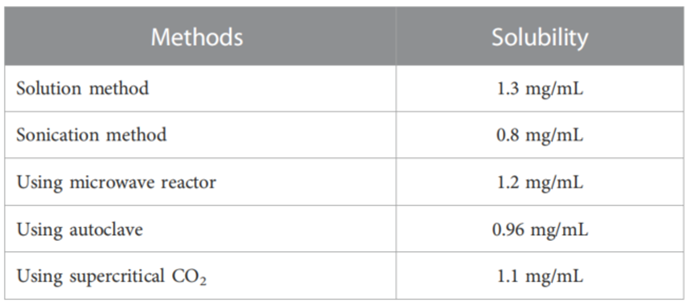
Tabel 1. The water solubilities of icariin in the complex from different methods.
3. In Vivo Pharmacokinetic Study
Procedure: Twelve healthy adult dogs were divided into two groups, receiving either icariin or the icariin-HP-γ-cyclodextrin complex orally at a dose of 20 mg/mL. Blood samples were collected at specified time points post-administration. Plasma samples were processed and analyzed using HPLC to measure icariin concentrations.
Result: The Cmax for the icariin complex group was found to be 9.06 μg/mL, while the Cmax for the icariin group was 1.84 μg/mL. The AUC0-120 values were significantly higher for the complex group (213.5 μg h/mL) compared to the icariin group (10.69 μg h/mL).
Finding: The pharmacokinetic analysis indicated that complexation with HP-γ-cyclodextrin substantially improved the bioavailability of icariin, evidenced by a fivefold increase in Cmax and a twentyfold increase in AUC0-120, suggesting enhanced therapeutic efficacy.
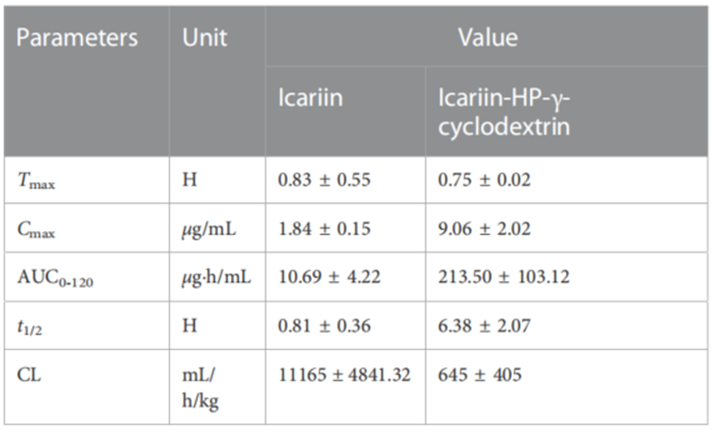
Table 2. Pharmacokinetic parameters of icariin and its HP-γ-cyclodextrin complex.
Enhanced Solubility and Pharmacokinetic Parameters of the Icariin Complex
This study presents a significant advancement in enhancing the solubility and pharmacokinetic profile of icariin, a natural hepatoprotector derived from Herba epimedii. The innovative complexation of icariin with hydroxypropyl-γ-cyclodextrin (HP-γ-cyclodextrin) resulted in an unprecedented 654-fold increase in water solubility, the highest recorded for icariin to date. Employing a combination of methodologies—such as optimizing the complexation conditions and utilizing various preparation techniques—this research successfully demonstrated that the inclusion complex not only improved the dissolution rate significantly but also facilitated a nearly 20-fold increase in bioavailability. These findings underscore the potential of HP-γ-cyclodextrin as an effective carrier for enhancing the therapeutic efficacy of poorly soluble drugs like icariin.
In conclusion, the research confirmed that the icariin-HP-γ-cyclodextrin complex exhibits a dramatic enhancement in pharmacokinetic parameters, including a fivefold increase in maximum concentration (Cmax) and an approximately twentyfold increase in the area under the curve (AUC0-120). The complexation reduced the clearance of icariin and extended its half-life, indicating that less drug is needed for the same therapeutic effect. These results suggest that the icariin complex could serve as a potent hepatoprotector or anti-liver cancer agent. This study provides a promising new formulation strategy that could lead to more effective treatments for liver-related health issues, thereby contributing to improved patient outcomes in hepatoprotection and cancer therapy.
Reference:
Ding, Yili, et al. “Improvement of solubility and pharmacokinetic profile of hepatoprotector icariin through complexation with HP-γ-cyclodextrin.” Frontiers in pharmacology 14 (2023): 1138686.
