Editor: Nina
Scientists develop Amphotericin B nano-assemblies using mango pulp extract to significantly reduce toxicity and enhance therapeutic efficacy against visceral leishmaniasis.
Key Preview
Research Question
The study primarily addresses the challenge of high intrinsic toxicity associated with the conventional use of Amphotericin B (AmB) in treating visceral leishmaniasis and other fungal infections. It investigates whether a novel formulation of AmB, in the form of nano-assemblies (AmB-NA), can effectively reduce toxicity while maintaining or enhancing therapeutic efficacy against Leishmania donovani.
Research Design and Strategy
The researchers employed a green synthesis approach to create AmB-NA, which involves using Mangifera indica (mango) pulp extract. This method aims to enhance drug delivery and efficacy while reducing the toxic side effects typically associated with AmB. The study design includes both in vitro and in vivo experiments to evaluate the pharmacokinetics, therapeutic efficacy, and toxicity profiles of the new formulation compared to conventional forms of AmB.
Method
The study utilized a series of experimental procedures, including the synthesis of nano-assemblies, in vitro toxicity assessments on red blood cells and macrophages, and in vivo evaluations of parasite burden in a BALB/c mouse model infected with L. donovani. Key measurements included the IC50 and CC50 values, evaluation of immune response, and analysis of biochemical parameters post-treatment.
Key Results
The results indicated that the AmB-NA formulation exhibited significantly lower toxicity compared to traditional AmB formulations, with less than 15% hemolysis at high concentrations. In vivo studies showed a substantial reduction in parasite load in the spleen, liver, and bone marrow of treated mice, with an IC50 value of 0.06 µM against amastigotes and 0.05 µM against promastigotes. The study also demonstrated robust Th1 immune polarization and a favorable safety profile, with minimal side effects.
Significance of the Research
This research presents a promising alternative to current Amphotericin B formulations, addressing the critical issue of toxicity in leishmaniasis treatment. The nano-assembled AmB formulation could lead to more effective and less toxic therapeutic options, enhancing patient compliance and treatment outcomes in visceral leishmaniasis.
Introduction
Visceral leishmaniasis (VL) is a severe parasitic disease caused by the protozoan parasite Leishmania donovani, primarily transmitted through the bites of infected sandflies. It represents a significant public health concern in many tropical and subtropical regions, especially in South Asia, where it poses a considerable morbidity and mortality risk. The clinical manifestations of VL include fever, weight loss, splenomegaly, and anemia, which can lead to life-threatening complications if left untreated. Current treatment options predominantly involve the use of drugs such as Amphotericin B (AmB), which is effective against VL but carries a high risk of toxicity.
Traditionally, drug delivery in the treatment of VL has relied on parenteral administration of conventional formulations of AmB, such as deoxycholate and liposomal preparations. These formulations are designed to enhance the solubility and bioavailability of the drug, allowing for effective targeting of the parasites within the host. However, despite their efficacy, these treatments present significant challenges, including severe side effects such as nephrotoxicity, infusion-related reactions, and the need for prolonged treatment regimens. These complications can result in poor patient compliance, increased healthcare costs, and ultimately, treatment failures. Moreover, the limited availability of certain formulations due to manufacturing constraints can restrict access to effective therapy, particularly in resource-limited settings.
In response to these challenges, innovative drug delivery strategies are being explored to enhance the therapeutic index of existing treatments. One promising approach is the development of nano-assembled formulations of AmB, which utilize biomimetic synthesis techniques to create stable nanoparticles with enhanced drug delivery properties. This strategy aims to reduce the toxicity associated with conventional formulations while maintaining or improving the drug’s efficacy against Leishmania donovani. By leveraging the unique properties of nanotechnology, these formulations offer the potential for controlled and sustained release of the drug, targeted delivery, and minimized side effects, ultimately leading to improved treatment outcomes for patients suffering from visceral leishmaniasis.
Research Team and Aim
The research team was led by Dr. Mohammad Owais, an expert in the field of biomedical research at Aligarh Muslim University. The study was conducted between November 2022 and December 2022, with the collaborative efforts of team members, including Fauzia Jamal, Ishrat Altaf, Ghufran Ahmed, and several others from both Aligarh Muslim University and the Rajendra Memorial Research Institute of Medical Sciences. The findings were published in the paper titled “Amphotericin B Nano-Assemblies Circumvent Intrinsic Toxicity and Ensure Superior Protection in Experimental Visceral Leishmaniasis with Feeble Toxic Manifestation,” appearing in the journal Vaccines.
The aim of the research, as articulated by Dr. Owais, was to develop a new formulation of Amphotericin B that maintains its therapeutic efficacy while significantly reducing toxicity, thereby improving treatment options for visceral leishmaniasis. This innovative approach seeks to address the critical challenges associated with current treatment strategies and enhance patient outcomes.
Experimental Process
Primary Technique: Green Synthesis of Amphotericin B Nano-Assemblies (AmB-NA)
The primary technique employed in this study is the green synthesis of Amphotericin B nano-assemblies (AmB-NA) using Mangifera indica (mango) pulp extract as a bio-reducing agent. This eco-friendly approach facilitates the formation of nano-sized drug delivery systems that enhance the therapeutic efficacy of Amphotericin B while minimizing toxicity.
Experiment 1: Preparation of Mangifera Indica Fruit Extract
Key Steps:
- Fresh Mangifera indica fruits were thoroughly washed to remove surface contaminants.
- The fruit pulp (30 g) was cut into small pieces and homogenized with 100 mL of MilliQ water to create a pulp mixture.
- The mixture was boiled for 10 minutes to extract bioactive compounds.
- The cooled mixture was filtered using Whatman filter paper, and the resultant extract was stored at -20 °C until further use.
Data Collection and Analysis:
The quantity of bioactive compounds in the mango pulp extract was not quantitatively measured in this step, but qualitative analysis was performed based on visual changes and the ability to reduce metal ions during the synthesis of nanoparticles.
Result:
The preparation of mango pulp extract provided a stable and rich source of phytochemicals, which were instrumental in the subsequent synthesis of AmB-NA.
Novel Aspects:
Using mango pulp extract as a reducing agent offers an environmentally friendly alternative to chemical synthesis methods, reducing the need for harmful chemicals.
Experiment 2: Synthesis of AmB-NA
Key Steps:
- A solution of Amphotericin B (1 mM in dimethyl sulfoxide) was prepared.
- A 5 mL aliquot of mango pulp extract was mixed with 5 mL of the Amphotericin B solution in a 10 mL volumetric flask.
- The mixture was stirred continuously at room temperature for 24 hours to allow for the formation of nano-assemblies.
- After 24 hours, the mixture was subjected to centrifugation at 20,000× g for 20 minutes at 4 °C to pellet down the formed nano-assemblies.
- The pellet was washed three times with phosphate-buffered saline (PBS) and resuspended in 5 mL PBS.
Data Collection and Analysis:
The size and morphology of the synthesized AmB-NA were characterized using Transmission Electron Microscopy (TEM) and Dynamic Light Scattering (DLS) techniques. The zeta potential was measured to assess the stability of the nanoparticles.
Result:
The AmB-NA exhibited an average size of approximately 97 ± 0.8 nm and a zeta potential of -34.2 ± 0.3 mV, indicating good stability.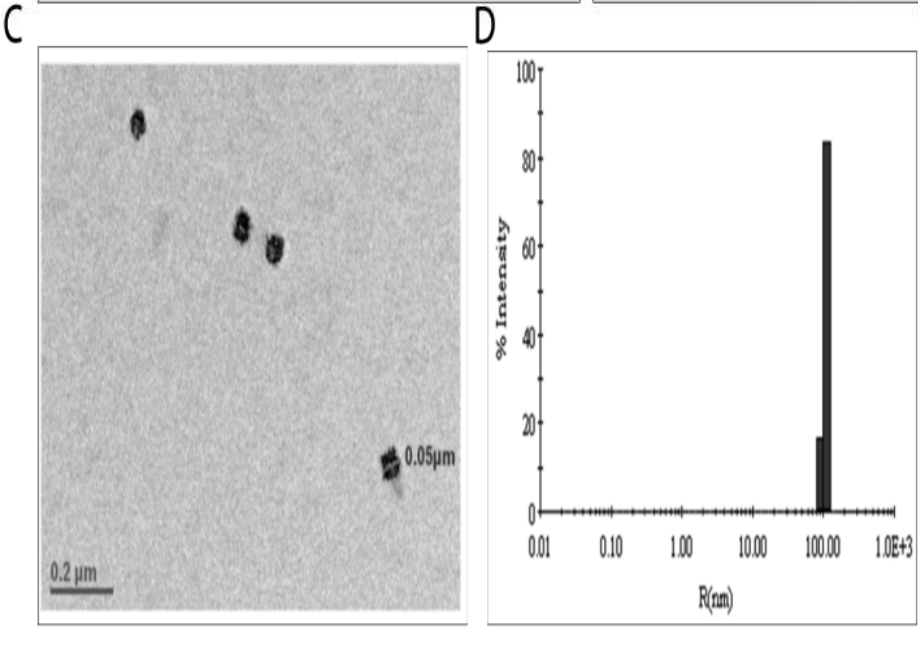
Figure 1. ©Size distribution and morphology of the as-synthesized AmB-NA as revealed by representative TEM image of the as-fabricated AmB-NA synthesized by emplloynig 5ml of Manglifera indica pulp extract mixed with 5 mL of 1 mM AmB solution. (D) the average diameter of AmB-NA,determined by DLS measurements was 97 ± 0.8 nm for AmB-NA.
Novel Aspects:
The method allows for the formation of stable nano-assemblies without the use of synthetic polymers or surfactants, addressing common drawbacks of traditional nano-delivery systems.
Experiment 3: In Vitro Drug Release Kinetics
Key Steps:
- A dialysis bag with a molecular weight cut-off of 12-14 kDa was used to assess the release kinetics of monomeric Amphotericin B from AmB-NA.
- 10 mL of AmB-NA solution (1 mg/2 mL) was placed in the dialysis bag, which was then submerged in 50 mL of PBS containing 5% DMSO.
- The setup was maintained at 37 °C on a magnetic stirrer.
- At designated time intervals (25 hours), 2 mL of sample was withdrawn from the PBS, and the absorbance of released Amphotericin B was measured at 408 nm using a UV-Vis spectrophotometer.
Data Collection and Analysis:
The amount of drug released was calculated and plotted against time to determine the release profile. The data were analyzed to determine the cumulative percentage of drug released over 250 hours.
Result:
The AmB-NA formulation demonstrated a sustained release profile, with approximately 46% of the drug released over 250 hours, indicating controlled drug release behavior.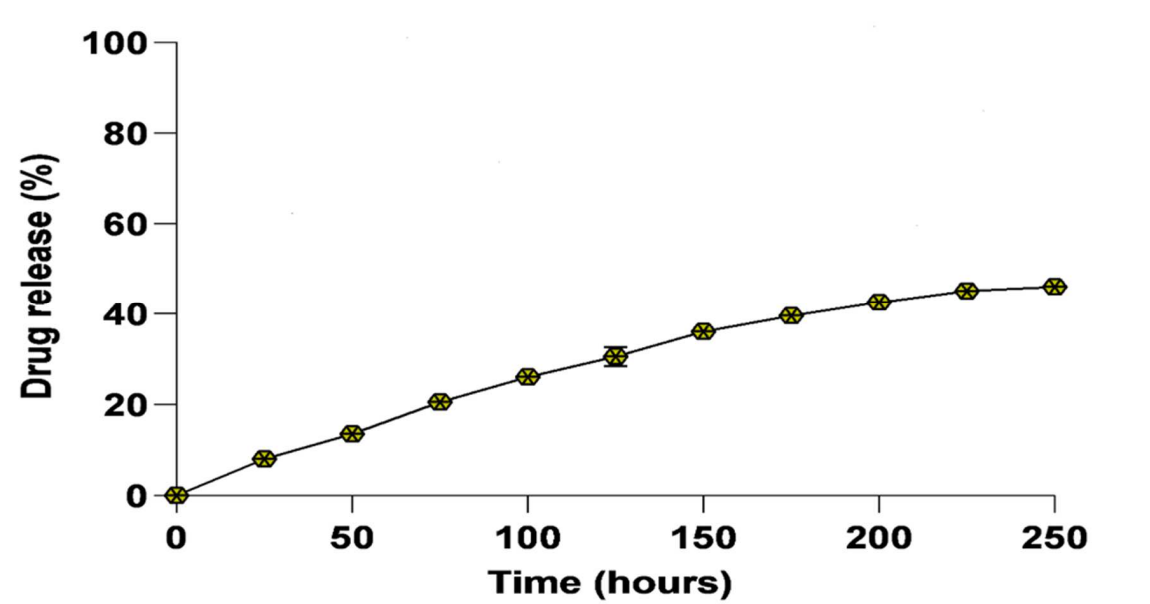
Figure 2. Release kinetics of AmB nano-assembly. Release kinetics of monomeric AmB from the assynthesized nano-assembled AmB in phosphate buffer saline (PBS) at pH 7.4 at 37 °C. The AmB released at different time periods was evaluated at 408 nm using a UV-Vis spectrophotometer. Each point depicts an average of three estimations ± SD.
Novel Aspects:
The sustained release kinetics of AmB-NA represent a significant improvement over traditional formulations, which often exhibit rapid drug release leading to higher toxicity.
Experiment 4: Toxicity Assessment
Key Steps:
- The hemolytic effect of AmB-NA on red blood cells (RBCs) was evaluated by incubating isolated RBCs with increasing concentrations of AmB-NA.
- Control groups included RBCs treated with Triton X-100 (100% lysis) and Fungizone™.
- After 24 hours of incubation, the samples were centrifuged, and the absorbance of the supernatant was measured at 576 nm to determine the percentage of hemolysis.
Data Collection and Analysis:
The absorbance readings were used to calculate the percent hemolysis of RBCs, providing a quantitative measure of AmB-NA toxicity.
Result:
AmB-NA exhibited less than 15% hemolysis at a concentration of 100 µg/mL, significantly lower than Fungizone™ and pure Amphotericin B, which displayed over 90% hemolysis.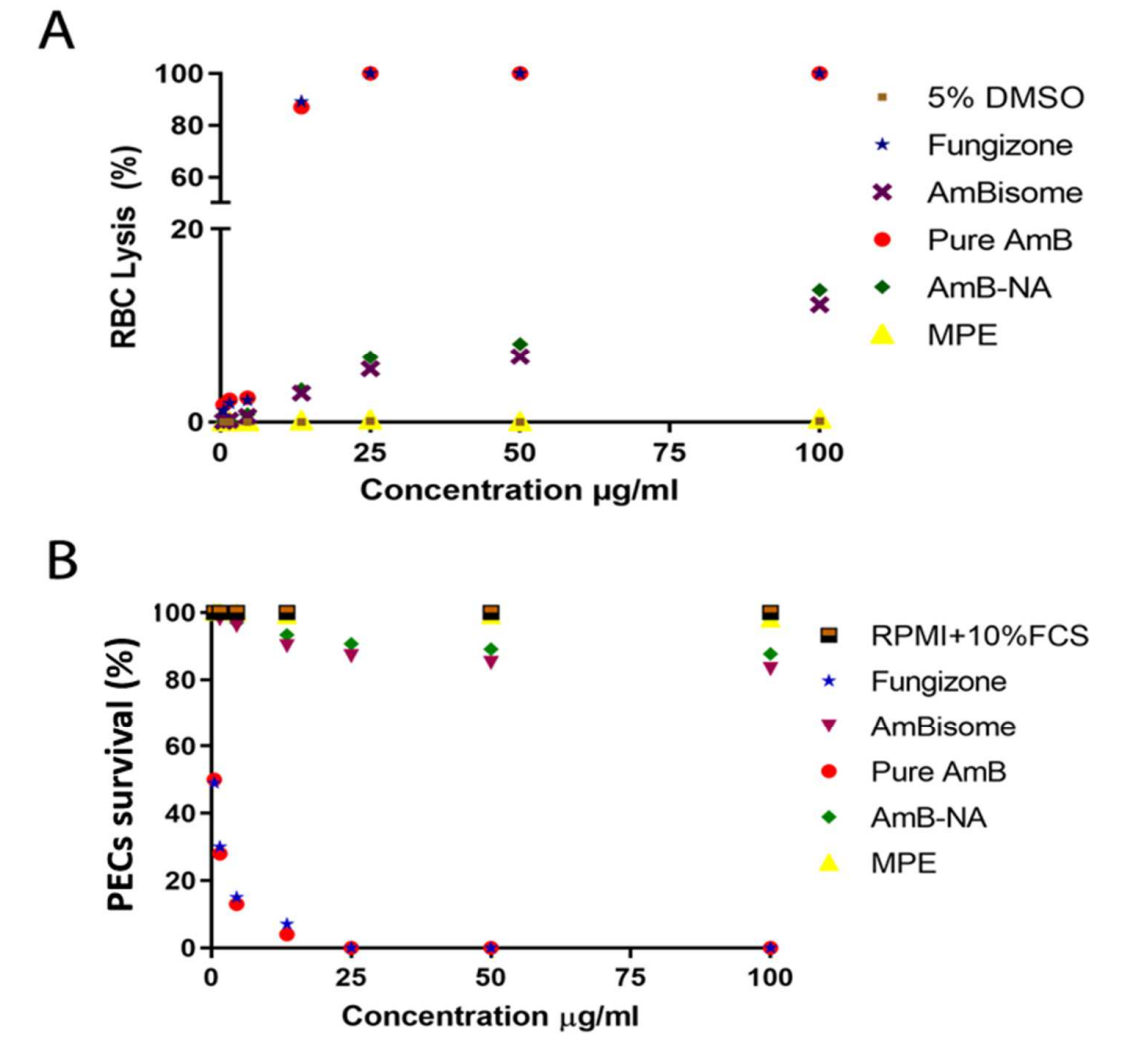
Figure 3. As-synthesized AmB-NA evoked minimal toxicity against living cells.
Novel Aspects:
The significantly lower hemolytic activity of AmB-NA compared to traditional formulations highlights its potential as a safer alternative in clinical applications.
Experiment 5: In Vivo Efficacy Studies
Key Steps:
- Six to eight-week-old BALB/c mice were infected with Leishmania donovani parasites.
- Different treatment groups received either saline (control), AmB, AmBisome, or AmB-NA (single and double doses).
- Post-treatment, the mice were sacrificed, and the spleen, liver, and bone marrow were harvested for parasite load assessment through Giemsa staining.
Data Collection and Analysis:
The number of amastigotes per 1000 cells was counted under a microscope, and the results were statistically analyzed to compare parasite burdens across treatment groups.
Result:
AmB-NA treatment resulted in a significant reduction in parasite loads in the spleen, liver, and bone marrow compared to control groups, demonstrating its efficacy.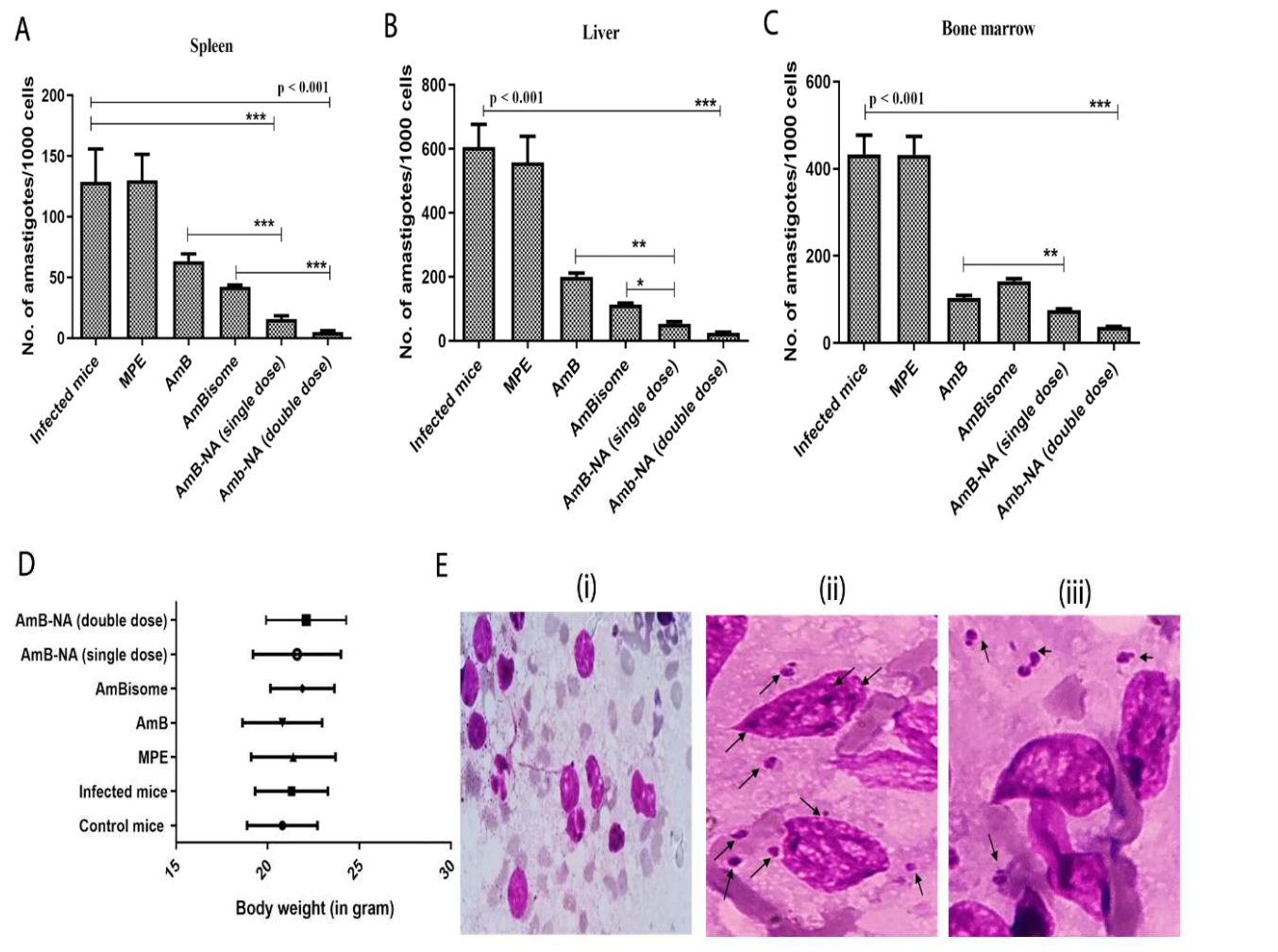
Figure 4. As-synthesized AmB-NA treatment resulted in successful clearance of the in vivo parasite burden. The treatment with AmB-NA resulted in the killing of the L. donovani systemic infection present in the experimental mice’s organs such as the spleen,liver,bone marrow etc. Infected mice with no AmB treatment were considered as controls.
Novel Aspects:
The use of AmB-NA led to superior parasite clearance compared to both free Amphotericin B and AmBisome, indicating a marked improvement in therapeutic outcomes.
Conclusion
The successful development of this drug delivery system, Amphotericin B Nano-Assemblies (AmB-NA), was achieved through innovative green synthesis techniques that utilized Mangifera indica (mango) pulp extract. This novel approach not only enhanced the physicochemical properties of Amphotericin B but also significantly reduced its intrinsic toxicity while maintaining strong anti-leishmanial efficacy.
The highlights of the study include the demonstration that AmB-NA exhibited a sustained drug release profile, effectively reduced parasite load in vital organs of experimental animals, and showed minimal toxicity compared to conventional formulations of Amphotericin B. Furthermore, the research provided compelling evidence of the formulation’s ability to induce a robust immune response, characterized by a shift towards a Th1 polarization, which is crucial for combating leishmaniasis. Overall, the findings suggest that AmB-NA presents a promising alternative to traditional Amphotericin B formulations, with the potential to improve treatment outcomes for patients suffering from visceral leishmaniasis.
Reference
Jamal, Fauzia, et al. “Amphotericin B Nano-Assemblies Circumvent Intrinsic Toxicity and Ensure Superior Protection in Experimental Visceral Leishmaniasis with Feeble Toxic Manifestation.” Vaccines, vol. 11, no. 1, 2023, p. 100. MDPI, https://doi.org/10.3390/vaccines11010100.
