Editor: Tiffany
A new study reveals that silencing miR-21 in sensory neurons reduces neuropathic pain by promoting an anti-inflammatory phenotype in macrophages, offering a potential new therapeutic target for this debilitating condition.
Key Highlights
- Research Question:
What is the role of miR-21 in sensory neurons in the development of neuropathic pain, and how does it interact with macrophages to influence pain mechanisms? - Research Difficulties:
Understanding the complex molecular mechanisms underlying neuropathic pain and identifying specific targets for therapeutic intervention without causing adverse effects. - Key Findings:
Silencing miR-21 in sensory neurons reduces neuropathic pain, decreases macrophage infiltration in the dorsal root ganglia, and promotes an anti-inflammatory phenotype in macrophages by activating the TGF-β pathway. - Innovative Aspects:
The study identifies a novel mechanism of neuron-macrophage communication involving miR-21 and the TGF-β pathway, providing new insights into the pathophysiology of neuropathic pain. - Importance of the Study:
The findings suggest that targeting miR-21 or the TGF-β pathway in macrophages could be a promising therapeutic strategy for neuropathic pain, potentially leading to more effective treatments for this condition.
Understanding Neuropathic Pain: A Persistent Clinical Challenge
Neuropathic pain is a chronic medical condition resulting from damage or dysfunction within the nervous system, leading to abnormal pain signaling. Unlike typical pain, which subsides as injuries heal, neuropathic pain persists, posing a significant clinical challenge due to its complexity. This condition arises from lesions or diseases affecting the somatosensory system, often manifesting as debilitating symptoms that severely impair patients’ quality of life.
The hallmark symptoms of neuropathic pain include chronic pain, hypersensitivity to non-painful stimuli (allodynia), and sensations such as tingling or burning. Patients may also experience spontaneous pain or an exaggerated response to painful stimuli (hyperalgesia). These symptoms stem from aberrant nerve signaling, making the condition notoriously difficult to manage.
Current therapeutic options for neuropathic pain include anticonvulsants like gabapentin, antidepressants such as amitriptyline, and, in severe cases, opioids. While these treatments can offer partial relief, their efficacy is limited, and they come with significant drawbacks. Side effects such as dizziness, sedation, and the potential for addiction are common, particularly with opioids. Moreover, these therapies primarily address symptoms rather than the underlying mechanisms driving the pain, underscoring the urgent need for innovative approaches to treatment.
Investigating miR-21’s Role in Neuropathic Pain Mechanisms
A study published on June 1, 2023, in The Journal of Clinical Investigation by Lynda Zeboudj and colleagues sought to unravel the molecular underpinnings of neuropathic pain. The research focused on microRNA-21 (miR-21), a small non-coding RNA molecule, and its role in sensory neurons and interactions with macrophages—immune cells implicated in pain modulation. The central research question was how miR-21 influences macrophage polarization and contributes to the development of neuropathic pain following nerve injury.
Investigating these mechanisms is fraught with challenges, as neuropathic pain involves a complex interplay of multiple cell types and signaling pathways. A primary difficulty lies in pinpointing specific molecular targets that can effectively modulate pain without triggering adverse effects. This study aimed to address these gaps by exploring miR-21 as a potential regulator of neuron-macrophage communication and its impact on pain hypersensitivity.
Experimental Approaches and Key Discoveries in miR-21 and Macrophage Interactions
Experimental Procedures
The research team employed a multifaceted approach to investigate miR-21’s role in neuropathic pain:
- Spared Nerve Injury (SNI) Model: Neuropathic pain was induced in mice using the SNI model, which mimics nerve injury conditions in humans.
- Conditional Knockout Mice: Mice lacking miR-21 in sensory neurons (miR-21 cKO) were generated to study the effects of miR-21 silencing.
- Behavioral Tests: Pain sensitivity was assessed using von Frey filaments, which measure mechanical allodynia by determining the force required to elicit a paw withdrawal response.
- Macrophage Analysis: Macrophage infiltration and phenotype in the dorsal root ganglia (DRG) were examined using flow cytometry and immunohistochemistry.
- Gene Expression Profiling: Microarray and quantitative PCR (qPCR) analyses were conducted on macrophages isolated from DRG to identify changes in gene expression.
- In Vitro Studies: Bone marrow-derived macrophages (BMDMs) were transfected with miR-21 mimics or antagomirs to manipulate miR-21 levels and assess downstream effects.
Key Experiments and Findings
1. Behavioral Tests in WT and miR-21 cKO Mice Post-SNI
- Procedure: Mechanical allodynia was measured using von Frey filaments at multiple time points following SNI in wild-type (WT) and miR-21 cKO mice.
- Result: At day 7 post-SNI, WT mice exhibited a paw withdrawal threshold of approximately 0.1 g, indicative of severe hypersensitivity. In contrast, miR-21 cKO mice showed a threshold of about 0.4 g, suggesting reduced pain sensitivity.
- Finding: Silencing miR-21 in sensory neurons significantly attenuates neuropathic pain, highlighting its critical role in pain development.
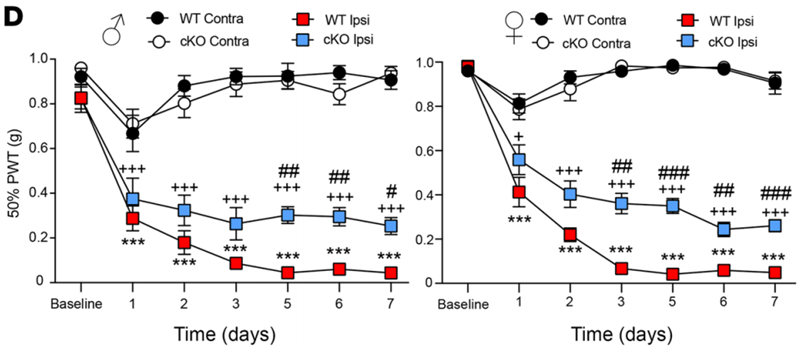
Figure 1. Attenuated allodynia in miR-21–cKO mice up to day 7 after SNI in males and females. Data are presented as 50% paw withdrawal thresholds (PWT).
2. Macrophage Infiltration in DRG
- Procedure: The presence of F4/80+ macrophages in the DRG was quantified via flow cytometry following SNI.
- Result: WT mice displayed a higher number of F4/80+ macrophages in the ipsilateral DRG compared to miR-21 cKO mice.
- Finding: Neuronal miR-21 promotes macrophage infiltration into the DRG after nerve injury, indicating its involvement in recruiting immune cells to the injury site.
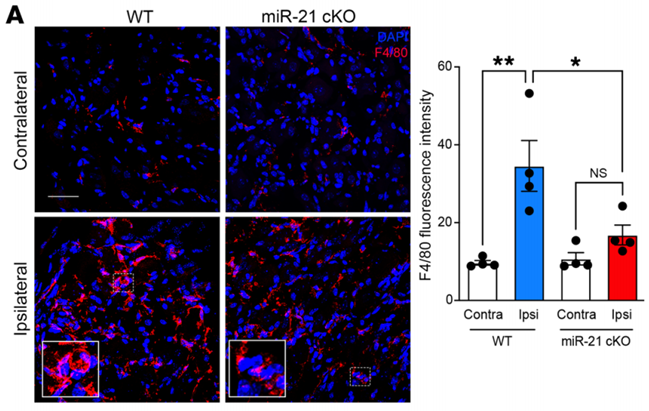
Figure 2. Immunofluorescent staining of F4/80 in ipsilateral and contralateral DRGs of WT and miR-21–cKO mice on day 7 after SNI.
3. Gene Expression Changes in Macrophages
- Procedure: Microarray analysis was performed on macrophages isolated from the DRG of WT and miR-21 cKO mice post-SNI.
- Result: Macrophages from miR-21 cKO mice showed a 3.02-fold increase in Tgfbr2 expression, alongside upregulation of other genes in the transforming growth factor-beta (TGF-β) pathway.
- Finding: The absence of neuronal miR-21 activates the TGF-β pathway in DRG macrophages, shifting them toward an anti-inflammatory phenotype.
4. In Vitro Studies with Macrophages
- Procedure: BMDMs were transfected with either a miR-21 mimic (to increase miR-21 levels) or an antagomir (to inhibit miR-21), followed by qPCR analysis of gene expression.
- Result: Mimic-21 transfection increased miR-21 levels by 240-fold, reduced Spry2 expression, and elevated pro-inflammatory markers such as Tnfa and Il6. Conversely, antagomir-21 transfection decreased miR-21, increased Tgfbr2, and reduced pro-inflammatory markers.
- Finding: miR-21 suppresses the TGF-β pathway and drives macrophages toward a pro-inflammatory (M1-like) phenotype, while its inhibition fosters an anti-inflammatory (M2-like) state.
Implications for Neuropathic Pain Treatment: Targeting miR-21 and Macrophage Polarization
This study reveals that silencing miR-21 in sensory neurons significantly reduces neuropathic pain by altering macrophage behavior in the DRG. Specifically, the absence of neuronal miR-21 prevents suppression of the TGF-β pathway in macrophages, enabling them to adopt an anti-inflammatory M2-like phenotype. This shift is associated with decreased pain hypersensitivity, as evidenced by higher paw withdrawal thresholds in miR-21 cKO mice compared to WT controls. Additionally, miR-21 promotes macrophage infiltration and pro-inflammatory responses, exacerbating pain following nerve injury.
The research uncovers a novel mechanism of neuron-macrophage communication mediated by miR-21 and the TGF-β pathway in the context of neuropathic pain. By demonstrating how miR-21 regulates macrophage polarization, the study identifies a promising therapeutic target for pain management. Current treatments fall short in addressing the root causes of neuropathic pain, but targeting miR-21 or enhancing TGF-β signaling in macrophages could pave the way for more effective strategies. These findings lay the groundwork for potential clinical trials of miR-21 inhibitors, offering hope for improved outcomes for patients suffering from this debilitating condition.
Reference:
Zeboudj, Lynda, et al. “Silencing miR-21-5p in sensory neurons reverses neuropathic allodynia via activation of TGF-β–related pathway in macrophages.” The Journal of clinical investigation 133.11 (2023).
