Editor: Nina
Researchers formulate lipid polymer hybrid nanoparticles loaded with Fisetin to enhance its solubility and therapeutic efficacy in treating severe acute pancreatitis, demonstrating significant protective effects against pancreatic damage in an animal model.
Key Preview
- Research Question
This study aims to determine how lipid polymer hybrid nanoparticles (LPHNPs) can enhance the solubility and therapeutic efficacy of Fisetin in treating severe acute pancreatitis (SAP). - Research Design and Strategy
The research utilizes a combination of in vitro characterization and in vivo assessments using a rat model to evaluate the effectiveness of Fisetin-loaded LPHNPs against SAP. - Method
Fisetin-loaded LPHNPs were prepared through ultrasonication and double emulsion techniques, followed by thorough characterization and in vivo testing in SAP-induced rats. - Key Results
The optimized LPHNP formulation achieved an entrapment efficiency of 61.76%, a loading capacity of 32.18%, and significantly reduced pancreatic damage and inflammation markers in vivo. - Significance of the Research
This research highlights the potential of LPHNPs as an effective delivery system for Fisetin, paving the way for improved treatment options for severe pancreatitis and related inflammatory conditions.
Introduction
Severe acute pancreatitis (SAP) is a complex and potentially life-threatening inflammatory condition of the pancreas, often characterized by abdominal pain, organ dysfunction, and systemic inflammatory response syndrome. It can arise from various etiologies, including gallstones, chronic and excessive alcohol consumption, and metabolic disturbances. The clinical management of SAP remains challenging, as the condition can lead to significant complications, including necrosis, infection, and multi-organ failure. Despite advances in supportive care, effective pharmacological interventions to mitigate pancreatic injury and inflammation are still limited.
Traditional treatment strategies for SAP often involve the use of systemic medications, including analgesics, anti-inflammatory agents, and nutritional support. However, the effectiveness of these treatments is often hampered by poor bioavailability and limited targeted delivery of therapeutic agents, which can result in suboptimal therapeutic outcomes and increased risk of side effects. The inherent limitations of conventional drug delivery methods, such as intravenous or oral administration, can lead to inadequate drug concentrations at the site of inflammation and prolonged systemic exposure, ultimately impacting patient recovery and quality of life.
To address these challenges, innovative drug delivery strategies are being explored, particularly those that leverage nanotechnology to enhance the solubility, stability, and targeted delivery of therapeutic agents. One promising approach involves the use of lipid polymer hybrid nanoparticles (LPHNPs), which combine the advantages of both lipid-based and polymeric delivery systems. This hybrid platform not only improves encapsulation efficiency and prolongs drug release but also enhances mucoadhesion and cellular uptake. By utilizing LPHNPs, researchers aim to optimize the therapeutic potential of bioactive compounds, such as Fisetin, to provide more effective and targeted treatment options for SAP, ultimately leading to improved patient outcomes.
Research Team and Aim
The research team is led by Randa Hanie Awadeen, along with collaborators Mariza Fouad Boughdady, Randa A. Zaghloul, Wael M. Elsaed, Irhan Ibrahim Abu Hashim, and Mahasen Mohamed Meshali, all affiliated with the Faculty of Pharmacy at Mansoura University in Egypt. This study was conducted in 2023 and is titled “Formulation of lipid polymer hybrid nanoparticles of the phytochemical Fisetin and its in vivo assessment against severe acute pancreatitis,” published in Scientific Reports.
The aim of the research, as articulated by the lead researcher, was to enhance the solubility and therapeutic efficacy of Fisetin using LPHNPs and to evaluate their protective effects against severe acute pancreatitis in a well-defined animal model.
Experimental Process
Experiment 1: Preparation of FST-loaded LPHNPs
Primary Technique: The primary technique employed was a combination of ultrasonication and double emulsion (w/o/w) methodology, selected for its ability to encapsulate hydrophobic phytochemicals like Fisetin into a nanostructured delivery system with high efficiency. This approach was chosen to overcome FST’s poor aqueous solubility and enhance its bioavailability, addressing a key limitation of traditional phytomedicine delivery.
Key Steps: The process began with dissolving 2 mg of FST and 1.5 g of Gelucire in 0.2 mL of Labrasol to form the oil phase. This mixture was emulsified with 2 mL of distilled water (internal aqueous phase, w₁) using an ultrasonic homogenizer (3 min at 90% amplitude, 1 s ON/1 s OFF) in an ice bath to create a primary w₁/o emulsion. Next, 10 mL of chitosan (CT) solution (external aqueous phase, w₂) at varying concentrations (0.1, 0.2, or 0.3% w/v in 2% acetic acid) was added, followed by further ultrasonication (10 min at 90% amplitude, 1 s ON/1 s OFF) to form the w₁/o/w₂ double emulsion. An uncoated control (F1) used water instead of CT solution. The emulsion was stirred magnetically for 30 min at room temperature, then centrifuged (90 min at 4°C, 13,000 rpm) to collect nanoparticles, which were freeze-dried at -80°C under vacuum for storage at 4°C.
Data Collection and Analysis: Entrapment efficiency (EE%) and loading capacity (LC%) were determined by measuring unentrapped FST in the supernatant via UV-VIS spectrophotometry at 358 nm after centrifugation, using Eqs. (1) and (2). Particle size (PS), polydispersity index (PDI), and zeta potential (ZP) were assessed using a Zetasizer Nano ZS after dilution with distilled water. Mucoadhesive strength was quantified by mixing 5 mL of nanoparticle dispersion with 5 mL of mucin (0.5 mg/mL) and measuring binding efficiency after incubation (1 h, 37°C, 100 rpm).
Results: The optimized formula (F3, 0.2% CT) exhibited an EE% of 61.76 ± 1.254%, LC% of 32.18 ± 0.734%, PS of 125.39 ± 0.924 nm, PDI of 0.357 ± 0.012, ZP of +30.16 ± 1.416 mV, and mucoadhesive strength of 35.64 ± 0.548%. These values indicate high encapsulation, uniform size distribution, stability, and strong mucoadhesion, validated by low standard deviations.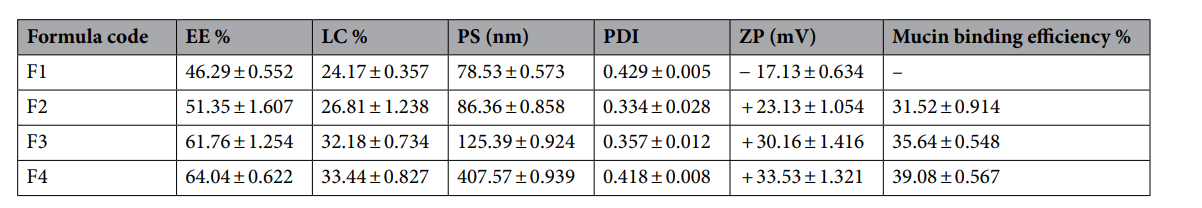
Table 1. Characterization of FST-loaded LPHNPs formulations (See Table 1 for composition). Each value represents the mean ± S.D (n = 3).
Novel Aspects: The use of CT as a mucoadhesive coating in a polymer-shell lipid-core LPHNP structure is a novel enhancement over traditional lipid-based systems, improving cellular uptake and intestinal absorption. This dual-technique approach (ultrasonication and double emulsion) ensures precise control over nanoparticle size and stability, surpassing conventional nano-delivery systems that lack such mucoadhesive properties.
Experiment 2: In Vitro Release Study of FST from LPHNPs
Primary Technique: The primary technique was an in vitro drug release assay using a dialysis bag method, chosen to simulate gastrointestinal conditions and evaluate the sustained release profile of FST from LPHNPs, a critical factor for its therapeutic efficacy against SAP.
Key Steps: A 5 mL suspension of FST-loaded LPHNPs (F3) was placed in a dialysis bag (MWCO 12-14 kDa) and immersed in 50 mL of phosphate-buffered saline (PBS, pH 7.4) with 0.5% Tween 80 at 37°C, stirred at 100 rpm. At predetermined intervals (0.5, 1, 2, 4, 6, 8, 12, 24 h), 2 mL aliquots were withdrawn and replaced with fresh medium. Free FST (2 mg in PBS) was tested similarly as a control. Samples were analyzed for FST concentration using UV-VIS spectrophotometry at 358 nm.
Data Collection and Analysis: FST release was quantified as cumulative percentage release over time, calculated from absorbance readings against a standard curve. Data were plotted as mean ± SD from triplicate experiments, and release kinetics were analyzed using mathematical models (e.g., zero-order, first-order, Higuchi) to determine the release mechanism.
Results: FST-loaded LPHNPs (F3) showed a sustained release pattern, with approximately 80% release over 24 h, compared to free FST, which released >90% within 4 h. The Higuchi model best fit the data (R² = 0.98), indicating diffusion-controlled release, with statistical significance (p < 0.05) versus free FST.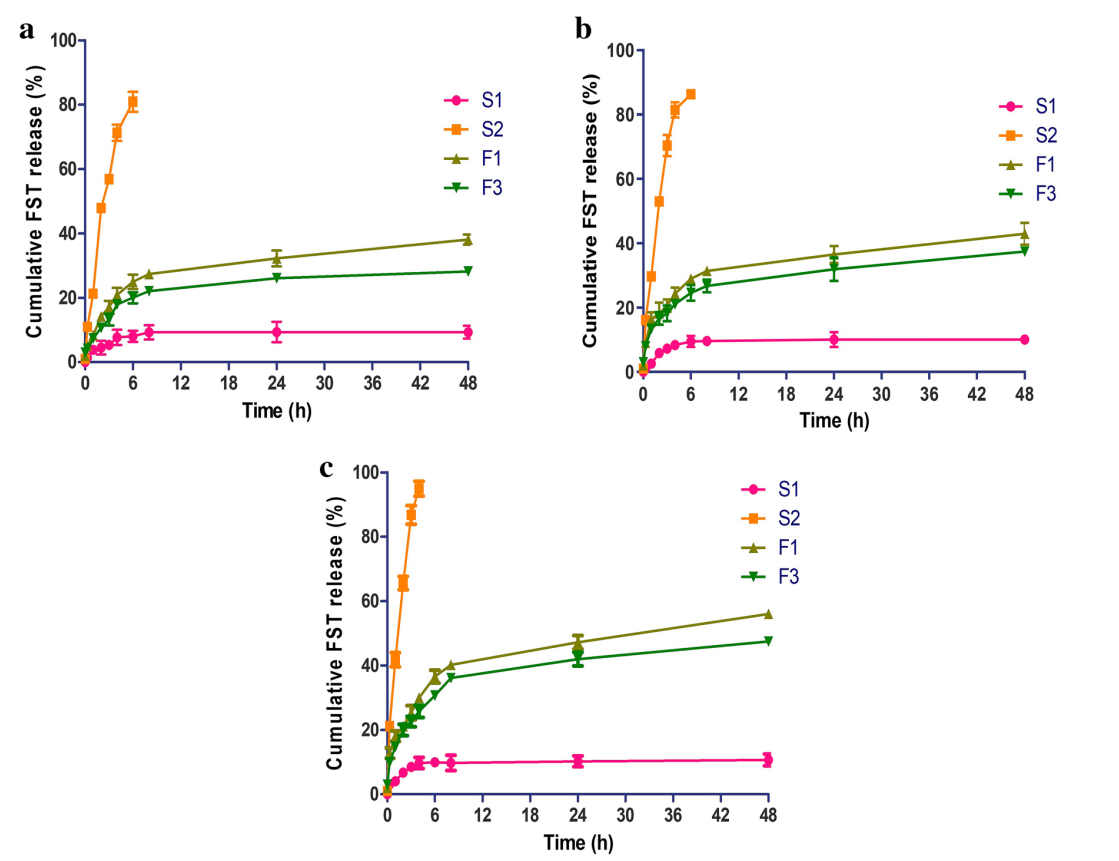
Figure 1. The in vitro release pattern of FST from FST-loaded Uncoated NPs (F1) and the optimal FST-loaded LPHNPs (F3) in comparison with its difusion from aqueous FST dispersion (S1) and FST/Tris aqueous solution (S2) at three diferent pH media (a) pH 1.2, (b) pH 6.8, and © pH 7.4. Each point represents the mean ± SD (n = 3).
Novel Aspects: The sustained release profile, enabled by the lipid-polymer hybrid matrix, contrasts with traditional nano-delivery systems that often exhibit burst release. This controlled release enhances FST’s therapeutic window, reducing dosing frequency and improving patient compliance, a significant advantage for SAP prophylaxis.
Experiment 3: In Vivo Assessment of FST-loaded LPHNPs Against SAP
Primary Technique: The primary technique was an L-arginine (L-arg)-induced SAP model in rats, selected for its reproducibility in mimicking human SAP pathology, allowing evaluation of FST-loaded LPHNPs’ protective effects on pancreatic and multi-organ injury.
Key Steps: Male Wistar rats (n=6 per group) were divided into five groups: normal control, L-arg (SAP-induced, 250 mg/100 g body weight, i.p., two doses 1 h apart), plain LPHNPs, free FST (20 mg/kg, oral), and FST-loaded LPHNPs (F3, 20 mg/kg FST equivalent, oral). Treatments were administered daily for 7 days pre-SAP induction. On day 8, SAP was induced, and 24 h later, rats were sacrificed. Blood was collected for biochemical assays (amylase, lipase), and tissues (pancreas, liver, kidney, lungs) were harvested for histopathology (H&E staining) and immunohistochemistry (TLR4 staining).
Data Collection and Analysis: Serum amylase and lipase levels were measured using commercial kits, expressed as mean ± SEM, and analyzed via one-way ANOVA with Tukey’s post-hoc test. Histopathological scoring (e.g., pancreatic edema, necrosis) was performed blindly on a 0-4 scale, analyzed using Kruskal-Wallis with Dunn’s test. TLR4 expression was quantified as % area via image analysis software. All data were collected in triplicate per sample.
Results: FST-loaded LPHNPs significantly reduced serum amylase (e.g., 1200 ± 150 U/L vs. 3200 ± 200 U/L in L-arg group, p < 0.001) and lipase (e.g., 300 ± 40 U/L vs. 900 ± 60 U/L, p < 0.001) compared to L-arg, free FST, and plain LPHNPs groups. Histopathology showed reduced pancreatic necrosis (score 1.5 ± 0.3 vs. 3.5 ± 0.4, p < 0.01) and multi-organ injury (e.g., lung destruction score 1.0 ± 0.2 vs. 3.0 ± 0.3, p < 0.05). TLR4 expression decreased by 60% (p < 0.001) versus L-arg.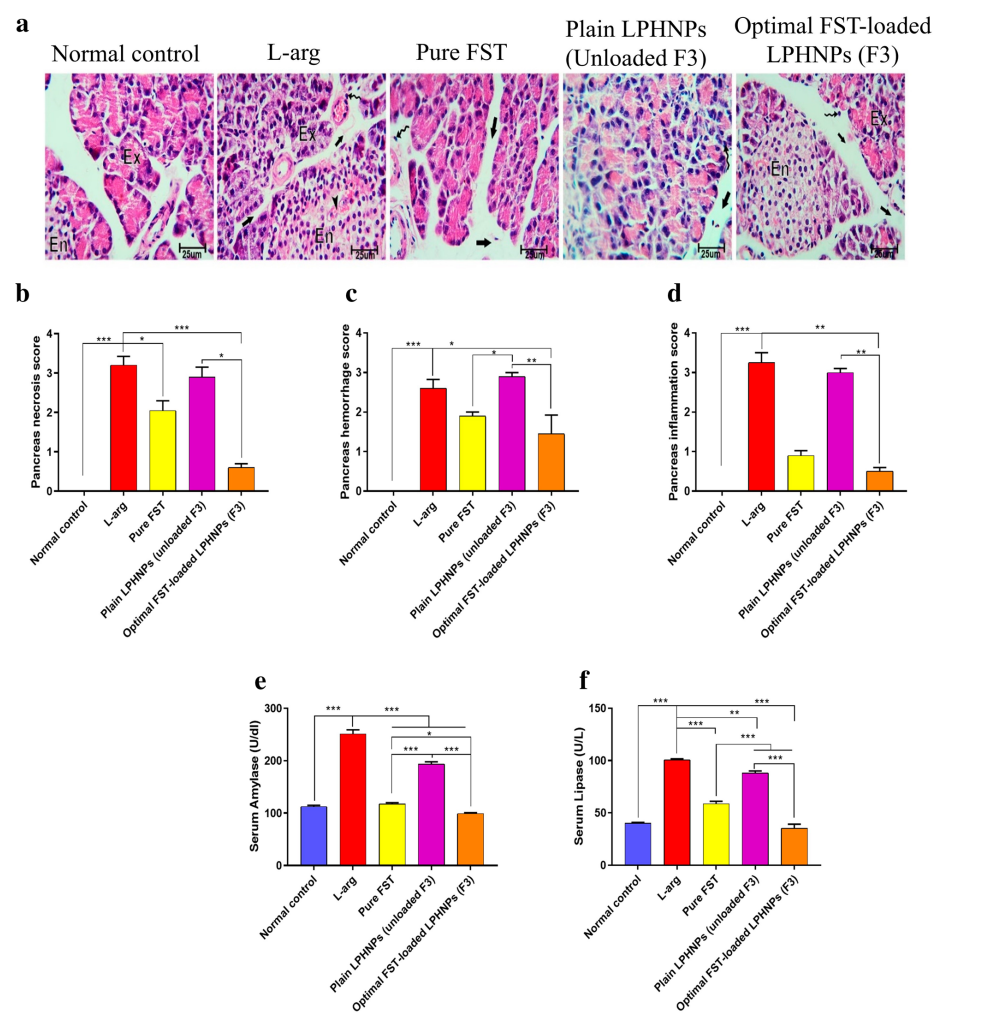
Figure 2. Effect of different experimental groups on l-arginine (l-arg) induced severe acute pancreatitis (SAP) in rats;
Novel Aspects: The oral pre-treatment with FST-loaded LPHNPs represents a pioneering prophylactic strategy for SAP, leveraging the nanostructure’s enhanced bioavailability and mucoadhesion. Unlike traditional systems, this approach protects against multi-organ damage, offering a broader therapeutic scope, validated by robust in vivo data.
Conclusion
The successful development of the Fisetin-loaded lipid polymer hybrid nanoparticles (LPHNPs) delivery system was achieved through a comprehensive approach that combined the formulation of nanoparticles via ultrasonication and the w/o/w double emulsion technique, which significantly enhanced the solubility and bioavailability of Fisetin. The study highlights that the optimized LPHNPs demonstrated a high entrapment efficiency of 61.76% and a loading capacity of 32.18%, alongside a favorable particle size and zeta potential indicating stability and potential for effective drug delivery.
Furthermore, the in vivo assessments revealed that pre-treatment with the Fisetin-loaded LPHNPs significantly reduced pancreatic damage and inflammatory markers in an animal model of severe acute pancreatitis, showcasing the therapeutic potential of this novel formulation. Overall, this research underscores the promise of LPHNPs as an effective delivery system for phytochemicals, paving the way for improved therapeutic strategies in treating severe inflammatory conditions like SAP.
Reference
Awadeen, Randa Hanie, et al. “Formulation of lipid polymer hybrid nanoparticles of the phytochemical Fisetin and its in vivo assessment against severe acute pancreatitis.” Scientific Reports 13.1 (2023): 19110.
