Editor: Nina
Scientists develop doxorubicin-loaded liposomal iron oxide nanoparticles for targeted chemo-photothermal cancer therapy, enhancing tumor accumulation and therapeutic efficacy through magnetic and laser-induced heating.
Key Preview
Research Question
This study investigates the development and efficacy of doxorubicin (DOX)-loaded liposomal iron oxide nanoparticles (Lipo-IONP/DOX) as a combined strategy for chemo-photothermal cancer therapy.
Research Design and Strategy
The research employs a novel formulation technique and evaluates the physicochemical properties, in vitro, and in vivo performance of Lipo-IONP/DOX. The approach focuses on enhancing drug delivery and therapeutic effectiveness through magnetic targeting and laser-induced heating.
Method
The synthesis of Lipo-IONP/DOX utilized a thin-film evaporation method, followed by characterization using various techniques including transmission electron microscopy (TEM), Fourier-transform infrared spectroscopy (FT-IR), and superconducting quantum interference device (SQUID) analysis. The in vivo efficacy was assessed in B16F10 melanoma-bearing mice.
Key Results
The study demonstrated that Lipo-IONP/DOX effectively accumulates in tumors through magnetic targeting, shows significant photothermal effects when irradiated, and enhances anti-cancer activity compared to free DOX alone. Tumor growth inhibition reached 84% in the combined treatment group.
Significance of the Research
This research presents an innovative strategy for improving the therapeutic efficacy of cancer treatments through targeted drug delivery and photothermal therapy, potentially leading to safer and more effective cancer treatments.
Introduction
Cancer remains one of the most formidable health challenges worldwide, characterized by uncontrolled cell proliferation and the potential for metastasis. Among its various forms, melanoma, a highly aggressive skin cancer, exemplifies the urgency for effective therapeutic interventions due to its rapid progression and resistance to conventional treatments. In 2023 alone, melanoma accounted for significant morbidity, underscoring the need for advanced strategies to improve patient outcomes.
Traditionally, cancer treatment relies heavily on systemic chemotherapy, where drugs such as doxorubicin (DOX) are administered intravenously to target rapidly dividing cancer cells. This approach utilizes the bloodstream to distribute chemotherapeutic agents throughout the body, aiming to reach tumor sites. While effective in some cases, this method depends on passive diffusion and the enhanced permeability and retention (EPR) effect to achieve sufficient drug concentrations at the tumor, a process often supplemented by liposomal formulations like Doxil to enhance stability and reduce toxicity.
However, the current systemic delivery approach faces distinct challenges. Poor tumor specificity results in low drug accumulation at the target site, with only a small fraction—typically less than 5% of the injected dose—reaching the tumor. This inefficiency leads to suboptimal therapeutic efficacy and necessitates higher doses, which exacerbate off-target effects, including cardiotoxicity and myelosuppression in the case of DOX. Additionally, the inability to confine therapeutic action to the tumor site limits the potential for localized enhancement of drug activity, often resulting in systemic toxicity and reduced patient quality of life.
To address these limitations, this study introduces an innovative drug delivery strategy: liposomal iron oxide nanoparticles loaded with doxorubicin (Lipo-IONP/DOX) for combined chemo-photothermal therapy. By integrating superparamagnetic iron oxide nanoparticles (IONPs) into a liposomal carrier, this approach leverages magnetic targeting to enhance tumor accumulation and near-infrared (NIR) photothermal therapy (PTT) to trigger localized drug release and induce hyperthermia. This dual-modality system aims to overcome the challenges of poor specificity and limited therapeutic confinement, offering a promising advancement in precision cancer treatment.
Research Team and Aim
This study was conducted by a collaborative research team led by Taehoon Park, a researcher from the College of Pharmacy and Research Institute of Pharmaceutical Sciences at Gyeongsang National University, Jinju, Republic of Korea. The research was carried out in late 2022, with the findings submitted for publication on November 27, 2022, and subsequently published on January 15, 2023. The team included contributors from both Gyeongsang National University and Inje University, specifically from the College of Pharmacy and Inje Institute of Pharmaceutical Sciences and Research in Gimhae, Republic of Korea. Their work was detailed in the paper titled “Liposomal Iron Oxide Nanoparticles Loaded with Doxorubicin for Combined Chemo-Photothermal Cancer Therapy,” published in the journal Pharmaceutics by MDPI.
The aim of the research, as articulated by Taehoon Park and colleagues, was to develop and evaluate a novel multifunctional nanoparticle platform: “Herein, we developed doxorubicin (DOX)-loaded liposomal IONP (Lipo-IONP/DOX) and evaluated in vitro and in vivo their applicability for combined chemo-photothermal cancer therapy.” This objective reflects the team’s intent to harness the unique properties of iron oxide nanoparticles (IONPs) and liposomes to create a targeted, dual-modality treatment system capable of enhancing cancer therapy through magnetic guidance and light-triggered drug release, ultimately improving therapeutic outcomes over traditional methods.
Experimental Process
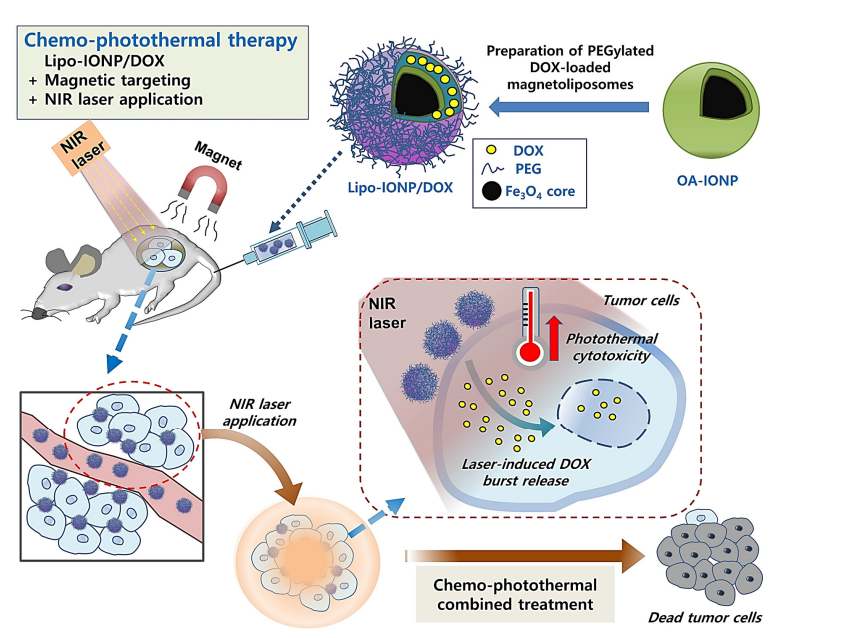
Experiment 1: Synthesis of Lipo-IONP/DOX
Primary Technique: The primary technique employed was the thin-film hydration method, a well-established approach for liposome preparation, chosen for its ability to encapsulate both hydrophobic iron oxide nanoparticles (IONPs) and hydrophilic doxorubicin (DOX) within a biocompatible lipid matrix.
Key Steps:
- Lipids (DPPC and DSPE-P2000 at a 5:1 molar ratio for L3 formulation) were dissolved in 1 mL chloroform with 1 mg Fe of oleic acid-coated IONP (OA-IONP).
- The mixture was incubated for 10 minutes at room temperature with shaking to ensure homogeneity.
- The solvent was evaporated under reduced pressure to form a thin lipid film.
- The film was hydrated with 1 mL pre-warmed (60°C) double-distilled water (DDW) or DOX solution (200 µg/mL) for 30 minutes.
- The resulting suspension was filtered through a 0.4 µm syringe filter and extruded against a 0.4 µm membrane to achieve uniform particle size.
Data Collection and Analysis: Particle size and zeta potential were measured using dynamic light scattering (DLS) with a Zetasizer Nano ZS. Iron content was quantified via inductively coupled plasma optical emission spectroscopy (ICP-OES). Lipid coating was confirmed by Fourier transform infrared (FT-IR) spectroscopy (400-4000 cm⁻¹).
Result: The L3 formulation yielded nanoparticles with a hydrodynamic size of 242.1 ± 1.2 nm, a zeta potential of -35.1 ± 0.2 mV, and an iron loading efficiency of 63.5 ± 6.4%, with DOX loading of 105 µg from a 200 µg feed (52.5% efficiency). These results confirmed successful encapsulation and stability.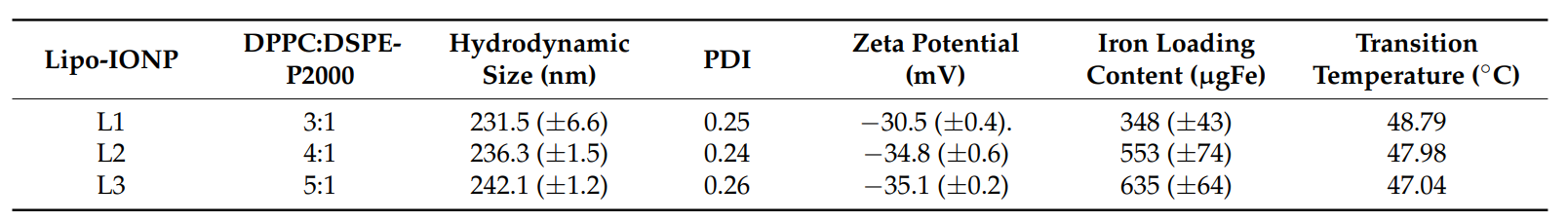
Table 1. Physical characterization of Lipo-IONP synthesized with different lipid ratios
Novel Aspects: Unlike traditional nano-delivery systems, this method integrates superparamagnetic IONPs and DOX into a single liposomal platform without stabilizers, leveraging the liposomes’ biocompatibility and the IONPs’ magnetic targeting capabilities, enhancing tumor-specific delivery over passive targeting systems.
Rationale: The thin-film hydration method was selected for its simplicity and ability to co-encapsulate hydrophobic and hydrophilic agents, addressing solubility challenges of IONPs and enabling dual-therapy functionality, critical for combined chemo-photothermal effects.
Experiment 2: Photothermal Activity Assessment
Primary Technique: Photothermal conversion was assessed using near-infrared (NIR) laser irradiation (885 nm), chosen for its deep tissue penetration and compatibility with IONP heat generation properties.
Key Steps:
- Lipo-IONP suspensions (150 µg Fe/mL) were prepared in 1 mL Eppendorf tubes.
- Samples were irradiated with a diode laser (MDL-III-885) at powers ranging from 0.7 to 1.5 W for 10 minutes.
- Additional samples at varying concentrations (25-200 µg Fe/mL) were irradiated at 1.3 W for 10 minutes.
- Photostability was tested with three cycles of 10-minute on/off laser exposure at 1.3 W.
Data Collection and Analysis: Temperature profiles were recorded using an infrared camera (FLIR E5). Data were plotted as temperature versus time or concentration, with maximum temperatures analyzed for consistency across cycles.
Result: At 150 µg Fe/mL and 1.3 W, the temperature reached 55°C, while 200 µg Fe/mL achieved 60°C. Photostability was confirmed with consistent peak temperatures (55°C) across three cycles, demonstrating robust heat conversion.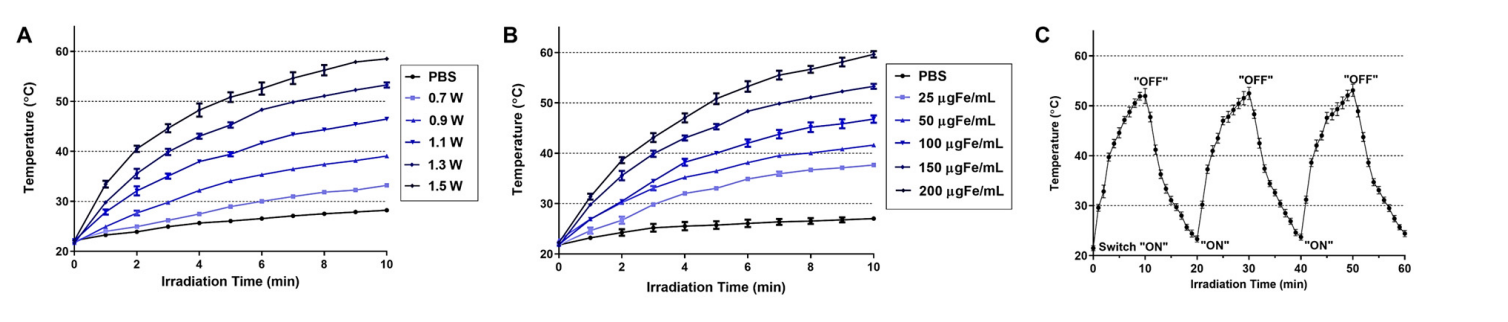
Figure 1. In vitro assessment of the light-to-heat conversion efficiency of Lipo-IONP. Temperature profiles of Lipo-IONP suspensions after laser irradiation (A) at different laser powers with fixed particle concentrations (150 µgFe/mL) and (B) at different particle concentrations with fixed laser power (1.3 W). © The stability of the Lipo-IONP’s photothermal activity was evaluated by monitoring the suspension temperature after laser irradiation for three cycles by a switch “on” and “off” mode (10 min for each mode in every cycle) (Lipo-IONP: liposomal iron oxide nanoparticle
Novel Aspects: The use of IONPs within liposomes provides superior photothermal stability compared to small molecule agents like indocyanine green, offering a reusable heat source for repeated therapy cycles, a limitation in traditional systems.
Rationale: NIR irradiation was chosen to maximize tissue penetration, and the cyclic testing ensured reliability for clinical translation, addressing heat confinement challenges in photothermal therapy (PTT).
Experiment 3: DOX Release Profile Evaluation
Primary Technique: Dialysis-based drug release testing was employed, selected for its ability to simulate physiological conditions and assess laser-triggered release kinetics.
Key Steps:
- Lipo-IONP/DOX (105 µg DOX loaded) was dispersed in 5 mL PBS (pH 7.4) in 20 mL glass vials.
- One group was irradiated with a 0.7 W laser for 10 minutes (reaching 48°C), while the control group was kept at room temperature.
- Samples were placed in dialysis bags (MWCO 14 kDa) and dialyzed against 50 mL PBS at 37°C with gentle agitation.
- At time points (0, 1, 2, 4, 6, 10, 24, 48 h), 1 mL aliquots were collected and replaced with fresh PBS.
Data Collection and Analysis: Released DOX was quantified by UV/VIS spectrophotometry at 490 nm. Cumulative release percentages were calculated and compared using Student’s t-test (p < 0.001).
Result: Laser-irradiated samples showed a burst release of 58.3% DOX at 24 hours versus 34.2% without irradiation, with both reaching 84% at 144 hours, confirming temperature-sensitive release at 48°C.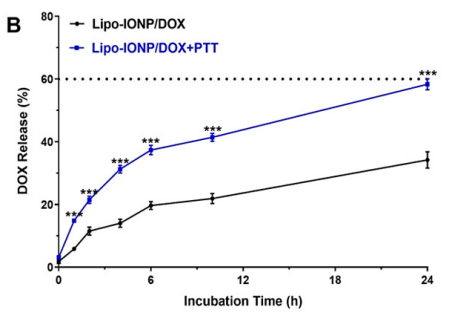
Figure 2. DOX release profiles with and without laser irradiation.
Novel Aspects: The thermosensitive liposome design enables on-demand DOX release triggered by PTT, a significant advancement over static release profiles in conventional nanocarriers, enhancing therapeutic precision.
Rationale: This method was used to mimic in vivo conditions and validate the transition temperature (47°C) of L3 liposomes, ensuring controlled drug release aligns with PTT application for synergistic effects.
Experiment 4: In Vitro Cytotoxicity Study
Primary Technique: The WST-1 cell viability assay was utilized, chosen for its sensitivity in quantifying cytotoxicity in B16F10 melanoma cells under combined chemo-photothermal conditions.
Key Steps:
- B16F10 cells were seeded in 96-well plates (5 × 10³ cells/well) and incubated overnight.
- Cells were treated with PBS, DOX, Lipo-IONP, or Lipo-IONP/DOX at DOX concentrations from 10⁻¹⁰ to 10⁻⁴ M.
- For photothermal testing, cells were treated with Lipo-IONP (150 µg Fe/mL) or Lipo-IONP/DOX (150 µg Fe/mL, 5 µM DOX) and irradiated at 0.9, 1.1, or 1.3 W for 10 minutes.
- After 48 hours, WST-1 reagent was added, and absorbance was measured at 450 nm.
Data Collection and Analysis: Cell viability was calculated relative to PBS controls. IC₅₀ values were determined, and photothermal effects were analyzed via 1-way ANOVA with Tukey’s post hoc test (p < 0.01).
Result: IC₅₀ values were 4.6 ± 0.8 µM (Lipo-IONP/DOX) and 6.1 ± 1.1 µM (DOX). With 1.3 W irradiation, Lipo-IONP/DOX reduced viability to 7.7%, significantly lower than DOX alone (p < 0.001), showing enhanced cytotoxicity.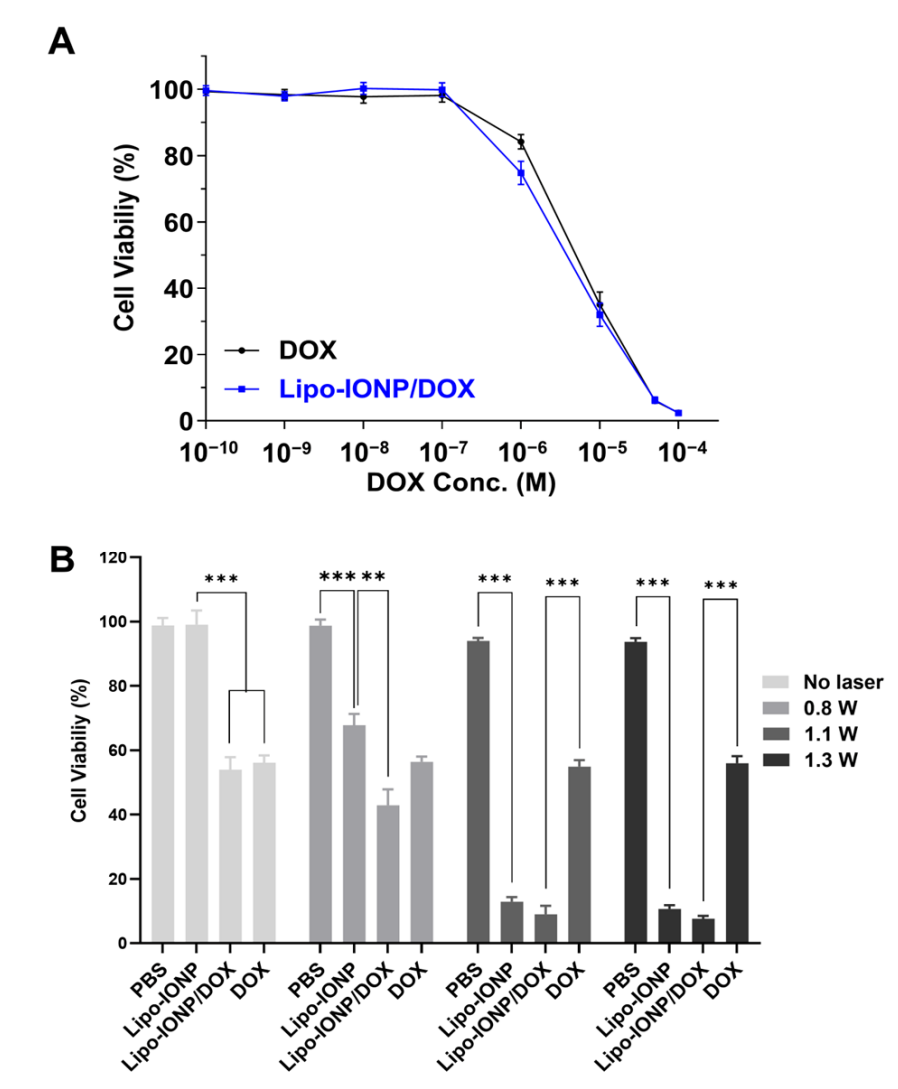
Figure 3. Cytotoxicity of Lipo-IONP/DOX.
Novel Aspects: Combining PTT with chemotherapy in a single nanoparticle outperforms standalone treatments, leveraging IONP heat generation to amplify DOX efficacy, unlike traditional separate delivery systems.
Rationale: This assay was chosen to directly compare chemo- and photothermal effects, validating the synergistic potential of Lipo-IONP/DOX in a controlled cellular environment.
Experiment 5: In Vivo Efficacy Study with Magnetic Targeting and PTT
Primary Technique: In vivo tumor growth inhibition was assessed in B16F10 subcutaneous tumor-bearing nude mice, selected to evaluate real-world therapeutic efficacy with magnetic targeting and PTT.
Key Steps:
- B16F10 cells (10⁷ cells/mouse) were injected subcutaneously into nude mice; tumors grew to 200 mm³ by day 9.
- Mice were divided into five groups (N=5): PBS, DOX (4 mg/kg), Lipo-IONP/DOX (24 mg Fe/kg, 4 mg/kg DOX), Lipo-IONP/DOX+MAG, and Lipo-IONP/DOX+MAG+PTT.
- For MAG groups, a 320 mT magnet was applied to tumors for 30 minutes post-injection.
- For PTT, tumors were irradiated with a 0.7 W laser (885 nm) for 10 minutes at 2 hours post-injection on days 9, 11, and 13.
- Tumor size was measured daily using calipers (V = (a² × b)/2).
Data Collection and Analysis: Tumor volumes were recorded until day 15 (PBS > 1500 mm³). Histological sections were stained with H&E and TUNEL, analyzed via fluorescence microscopy. Statistical significance was assessed by 1-way ANOVA (p < 0.001).
Result: Lipo-IONP/DOX+MAG+PTT inhibited tumor growth by 84% (290 mm³) versus PBS (1840 mm³), with significant apoptosis (TUNEL-positive cells) compared to 37% inhibition (1170 mm³) by Lipo-IONP/DOX alone (p < 0.001).
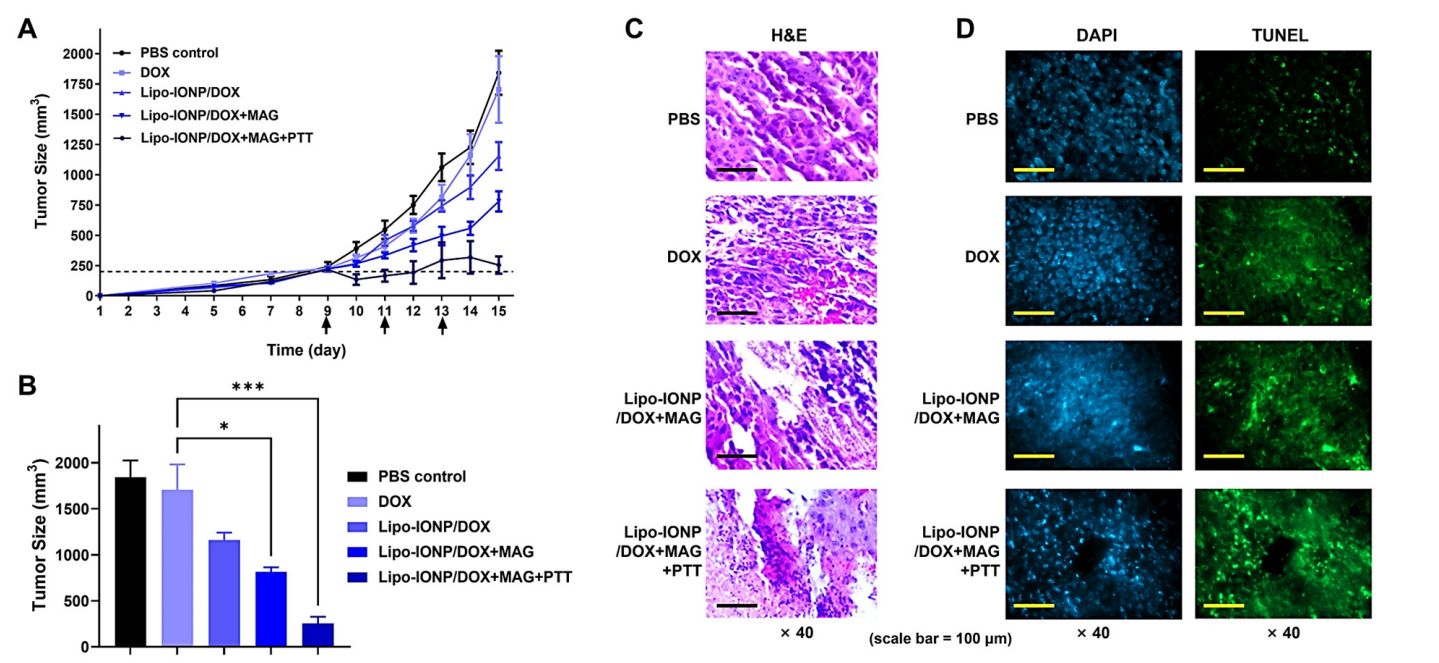
Figure 4. Efficacy study results of the combined chemo-photothermal therapy with Lipo-IONP/DOX in B16F10 s.c. allograft tumor mouse model.
Novel Aspects: Magnetic targeting increases tumor accumulation 6.3-fold (6.7% I.D./g), and PTT triggers DOX release, offering a multimodal approach superior to passive targeting or single-mode therapies in traditional systems.
Rationale: This model was chosen to replicate clinical tumor conditions, with magnetic targeting and PTT enhancing specificity and efficacy, addressing limitations of systemic delivery and heat confinement.
Conclusion
The successful development of the doxorubicin-loaded liposomal iron oxide nanoparticles (Lipo-IONP/DOX) as a combined chemo-photothermal cancer therapy system was achieved through a systematic approach that integrated innovative synthesis techniques, thorough characterization, and rigorous in vitro and in vivo evaluations. The thin-film evaporation method facilitated the efficient encapsulation of both the hydrophilic drug doxorubicin and the hydrophobic iron oxide nanoparticles, resulting in a stable and biocompatible formulation with a high drug loading efficiency of 63.5%.
The highlights of this study include the demonstration of significant tumor targeting capabilities through magnetic guidance, which resulted in a remarkable 6.3-fold increase in tumor accumulation of the Lipo-IONP/DOX compared to passive targeting. Additionally, the combination of photothermal therapy with chemotherapy showed enhanced cytotoxicity against B16F10 melanoma cells, achieving an impressive 84% tumor growth inhibition in vivo. These findings not only underscore the potential of Lipo-IONP/DOX as an effective therapeutic agent for cancer treatment but also pave the way for further research into multi-modal drug delivery systems that can improve patient outcomes while minimizing side effects associated with conventional therapies. Overall, this innovative approach highlights the promise of advanced drug delivery strategies in addressing the persistent challenges of cancer treatment.
Reference
Park, Taehoon, et al. “Liposomal iron oxide nanoparticles loaded with doxorubicin for combined chemo-photothermal cancer therapy.” Pharmaceutics 15.1 (2023): 292.
