Key Preview
- Research Question: Can a synergistic pH-responsive serum albumin-based drug delivery system improve the efficacy of doxorubicin and betulinic acid against non-small cell lung cancer?
- Research Design and Strategy: The study utilized an oil-water micro-emulsion method to encapsulate Dox and BeA within BSA nanoparticles.
- Method: The encapsulation efficiency, drug release profile, and cytotoxic effects were evaluated through a series of in vitro assays on A549 NSCLC cells.
- Key Results: The system demonstrated a high encapsulation efficiency (77% for Dox and 18% for BeA) and enhanced cytotoxic effects compared to the drugs alone, indicating a synergistic interaction.
- Significance of the Research: This study provides a promising strategy for improving the therapeutic effects of existing anticancer drugs while potentially reducing side effects and drug resistance.
Introduction
Non-small cell lung cancer (NSCLC) represents a significant health challenge, accounting for approximately 85% of all lung cancer cases and leading to high mortality rates worldwide. Characterized by its heterogeneous nature, NSCLC includes various subtypes, such as adenocarcinoma, squamous cell carcinoma, and large cell carcinoma. Early-stage detection often leads to better prognoses; however, the majority of patients are diagnosed at advanced stages, where treatment options become limited, and the disease becomes increasingly resistant to conventional therapies.
In traditional treatment strategies for NSCLC, systemic chemotherapy remains a cornerstone, utilizing agents such as doxorubicin (Dox) to inhibit cancer cell proliferation and induce apoptosis. However, the conventional delivery of these chemotherapeutic agents faces several limitations, including poor solubility, lack of specificity to tumor sites, and systemic toxicity. As a result, patients often experience severe side effects, and the efficacy of treatment diminishes over time due to the development of drug resistance. The non-specific nature of chemotherapy not only affects cancer cells but also harms surrounding healthy tissues, leading to adverse outcomes and reduced quality of life.
These challenges underscore the urgent need for innovative drug delivery strategies that can enhance the therapeutic index of existing anticancer agents while minimizing their toxic effects. A promising approach involves the development of nanosized drug delivery systems (DDS), which can improve drug solubility, stability, and bioavailability. These systems can be designed to facilitate targeted delivery, allowing for higher concentrations of drugs at tumor sites while sparing normal tissues.
This study introduces a novel pH-responsive drug delivery system utilizing bovine serum albumin (BSA) as a carrier for encapsulating both Dox and betulinic acid (BeA), a natural triterpene with demonstrated anticancer properties. By leveraging the unique properties of albumin and the tumor microenvironment’s acidic conditions, this innovative DDS aims to enhance the therapeutic efficacy of the drug combination while mitigating the challenges associated with traditional chemotherapy.
Research Team and Aim
The research was conducted by a multidisciplinary team led by Dr. Zally Torres-Martinez, who spearheaded the project in collaboration with colleagues from various institutions, including the University of Puerto Rico and San Juan Bautista School of Medicine. This collaborative effort culminated in the publication of their findings in the paper titled “A Synergistic pH-Responsive Serum Albumin-Based Drug Delivery System Loaded with Doxorubicin and Pentacyclic Triterpene Betulinic Acid for Potential Treatment of NSCLC,” which appeared in the journal BioTech.
The primary aim of the research, as articulated by Dr. Torres-Martinez, was to develop an effective drug delivery system that could improve the therapeutic outcomes of Dox and BeA in the treatment of non-small cell lung cancer (NSCLC). The team sought to leverage the unique properties of bovine serum albumin (BSA) as a carrier to enhance drug solubility and stability while providing a targeted approach to minimize systemic toxicity and overcome challenges related to chemoresistance.
Experimental Process
Primary Technique
The primary technique employed in this study was the oil-in-water micro-emulsion method for the preparation of bovine serum albumin (BSA)-based nanoparticles (NPs) encapsulating doxorubicin (Dox) and betulinic acid (BeA). This method was chosen for its ability to effectively encapsulate hydrophobic drugs within a biocompatible protein matrix, allowing for targeted delivery and enhanced therapeutic efficacy against non-small cell lung cancer (NSCLC).
Key Steps
- Preparation of Nanoparticles:
- BSA was dissolved in a 1× phosphate buffer solution at pH 7.4 to achieve a concentration of 5 mg/mL at 40 °C.
- Dox and BeA were separately dissolved in N,N-Dimethylformamide (DMF).
- The drug solution was added to the BSA solution using a syringe needle at a constant flow rate of 120 mL/h, maintaining a final protein-to-drug ratio of 1:2 (w:w).
- The emulsion was stirred at 100 rpm for 4 hours to allow for nanoparticle formation.
- Centrifugation and Filtration:
- The emulsion was subjected to centrifugation three times with nanopure water using a 10 kDa filter unit at 5000 rpm for 10 minutes to remove any unencapsulated drugs.
- The final nanoparticle solution was freeze-dried for 48 hours and stored at -20 °C until further use.
- Characterization of Nanoparticles:
- Dynamic light scattering (DLS) was used to measure the size, polydispersity index, and zeta potential of the nanoparticles, ensuring uniformity and stability.
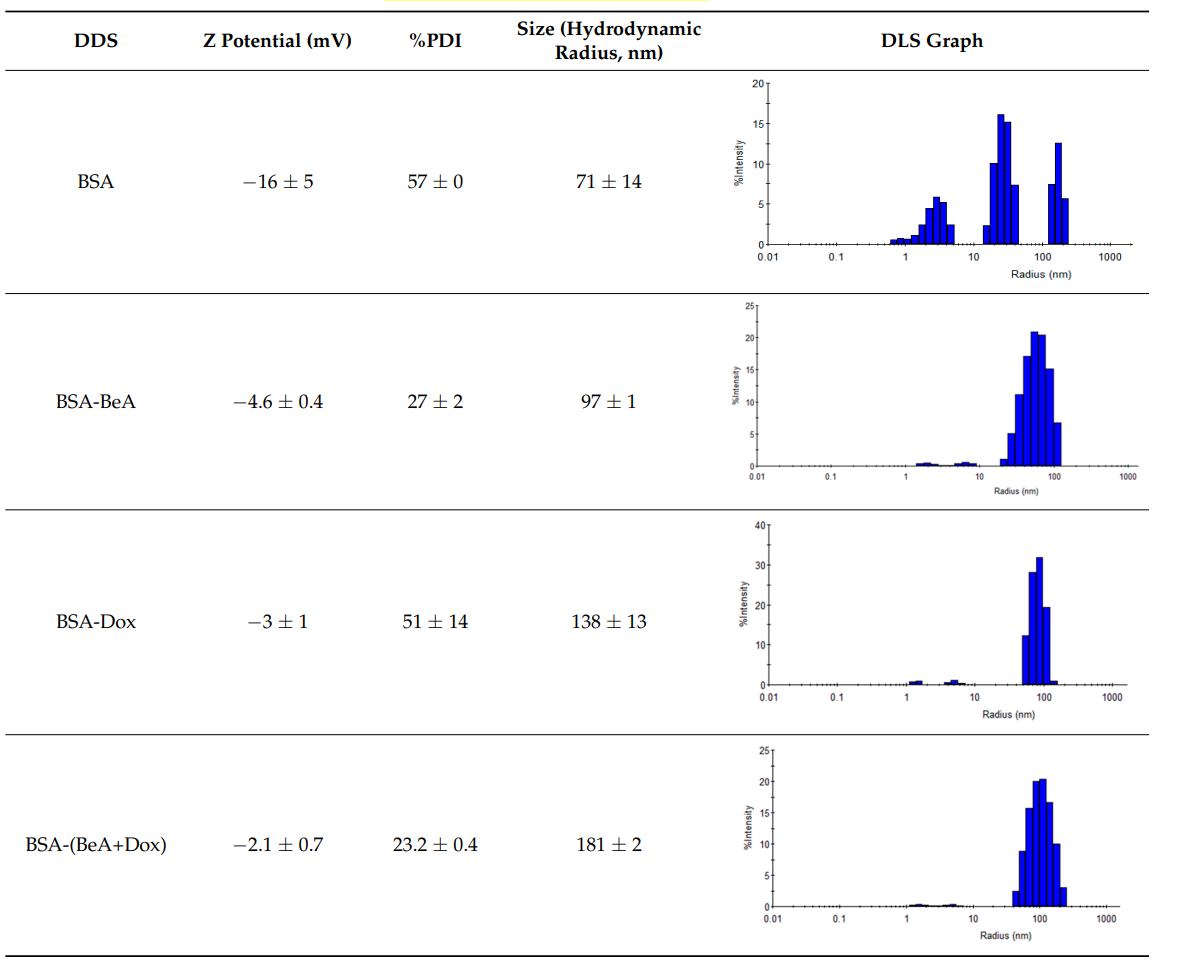
Table 1. Characterization of the protein-based drug delivery system by DLS
- Circular dichroism spectroscopy was performed to assess alterations in the secondary structure of BSA after drug loading, confirming successful encapsulation of Dox and BeA.
- In Vitro Drug Release Studies:
- Drug release profiles were evaluated by incubating the nanoparticles in sodium bicarbonate buffer at pH 6.8 (tumor-like conditions) and pH 7.4 (physiological conditions) at 37 °C.
- At predetermined intervals, samples were filtered, and the concentrations of released Dox and BeA were measured using spectrophotometric techniques, allowing for the determination of cumulative release over time.
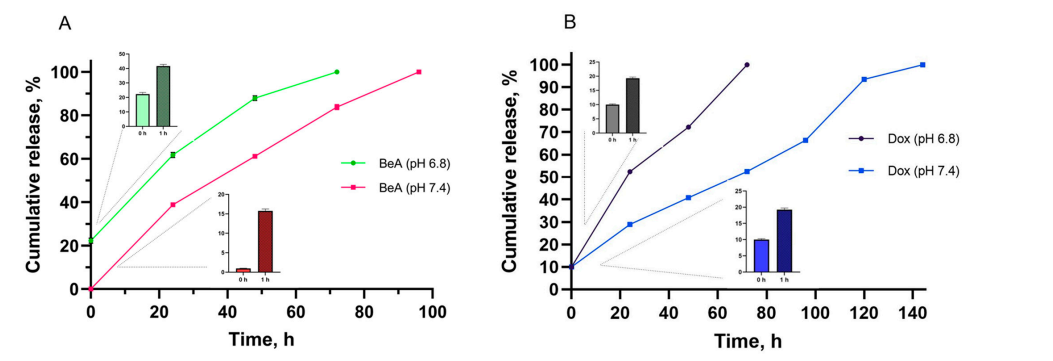
Figure 1. Cumulative in vitro release of the encapsulated drugs BeA (A) and Dox (B) from the DDS. The insets represent the initial burst release of each drug at 0 and 1 h insets. DDS was incubated in sodium bicarbonate buffer (pH 6.8 and 7.4) at 37 ◦C, maintaining sink conditions. All data are presented as mean ± SD
- Cytotoxicity and Synergistic Effect Analysis:
- A549 NSCLC cells were seeded in 96-well plates and treated with various concentrations of the BSA-(Dox+BeA) nanoparticles, Dox, and BeA alone for 24 hours.
- Cell viability was assessed using the MTS assay, and the combination index (CI) was calculated using CompuSyn software to evaluate the synergistic interactions between Dox and BeA.
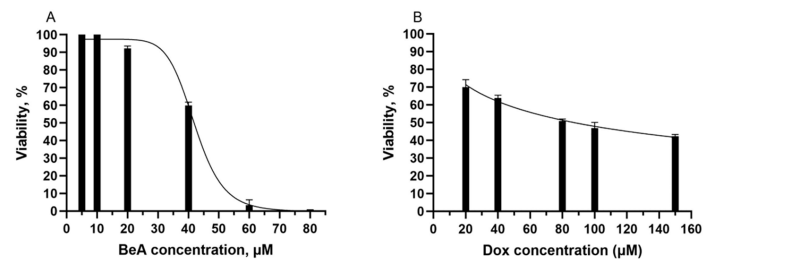
Figure 2. Viability of A549 cells incubated with different concentrations of BeA (A) and Dox (B) determined by MTS assay. All data are presented as mean ± SD
- Mechanistic Studies:
- Flow cytometry was utilized to analyze cell cycle distribution, caspase activation, DNA damage, oxidative stress, and EGFR expression in treated A549 cells, providing insights into the mechanisms underlying the cytotoxic effects of the nanoparticles.
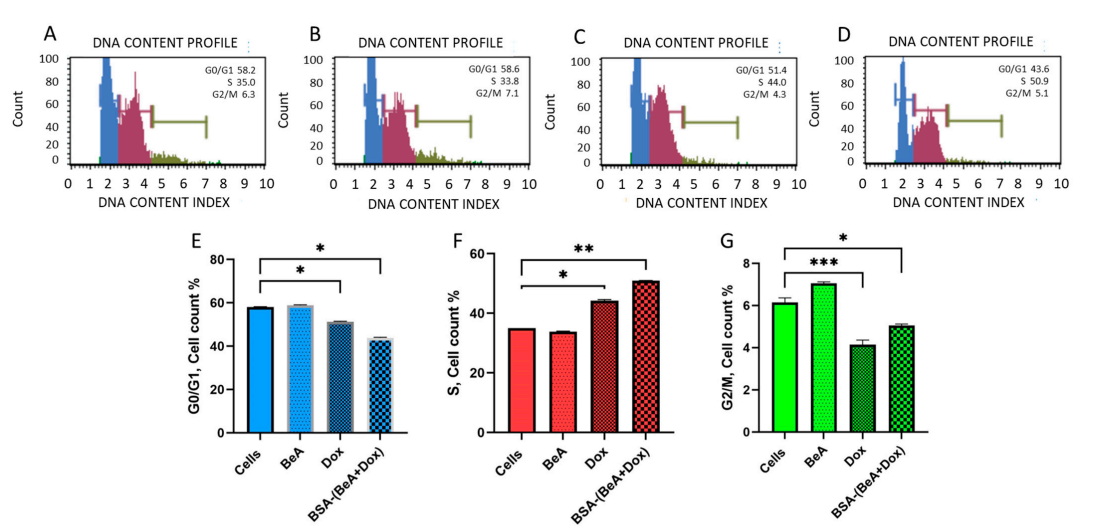
Figure 3. Cell cycle index induced by treatment of A549 cells with BSA-(BeA+Dox). The cell cycle distribution of untreated A549 cells (A) and cells treated with BeA (B), Dox ©, and BSA-(Bea + Dox) (D) is presented. Comparison of cell populations in G0/G1 (E), S (F), and G2/M (G) phases of A549 cells for each sample are shown in (A–D). Data are presented as mean ± SD with statistical significance. Significant differences between the means were accepted when the p-value was lower than 0.05 (), 0.01 (), and 0.001 ()
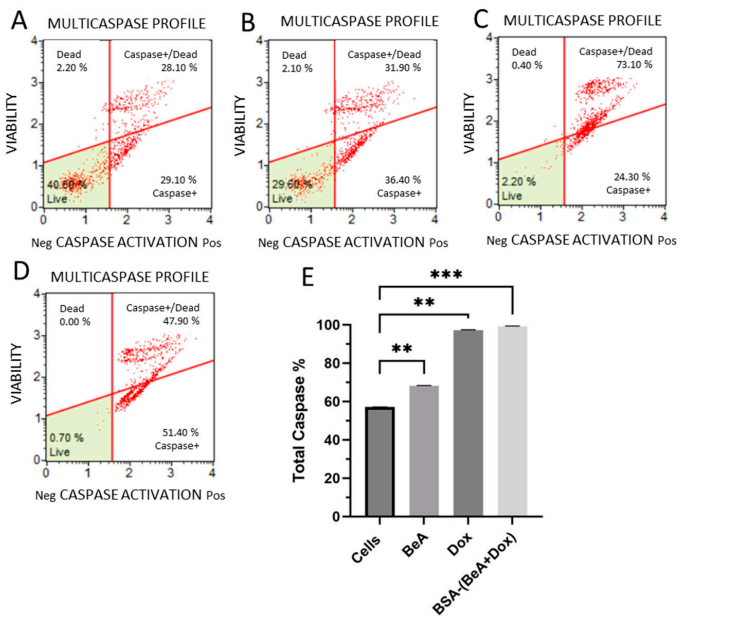
Figure 4. Activation of caspases by BSA-(BeA+Dox). The percentage and distribution of cells exhibiting caspase activity; (A) untreated A549 cells and cells treated with BeA (B), Dox ©, and BSA-(BeA+Dox) (D). Data are presented as the means ± SD (E) with statistical significance for all treated cells versus the control. Significant differences between the means were accepted when the p-value was lower than 0.01 (), and 0.001 (*).
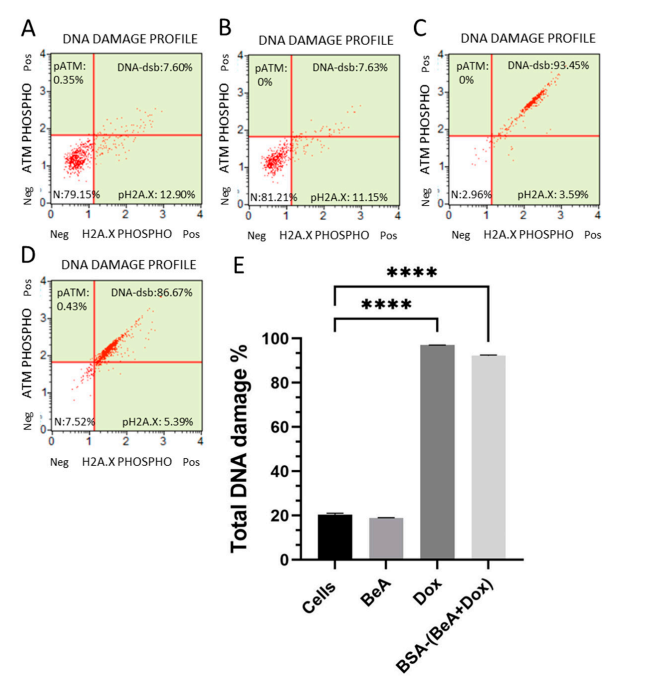
Figure 5. The distribution of DNA damage in untreated cells (A) and cells treated with BeA (B), Dox ©, and BSA-(BeA+Dox) (D) is presented. Data are presented as the means ± SD (E). Significant differences between the means were accepted when the p-value was lower than 0.0001 (****)
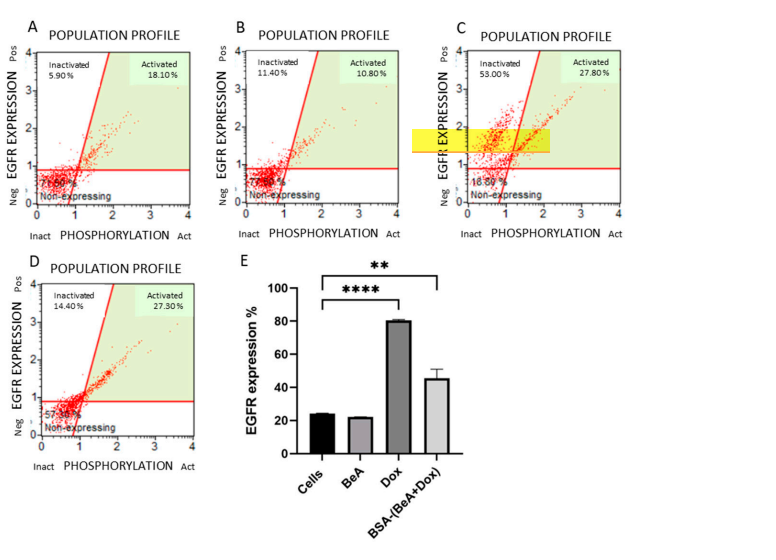
Figure 6. Determination of EGFR expression produced by treatment of A459 cells. Distribution of EGFR expression in untreated cells (A) and cells treated with BeA (B), Dox ©, and BSA-(BeA+Dox) (D). Data are presented as the means ± SD (E). Significant differences between the means were accepted when the p-value was lower than 0.01 (**), and 0.0001 (****)
Data Collection and Analysis
Data was collected through a series of quantitative assays, including spectrophotometric measurements for drug concentration and fluorescence imaging for cellular uptake studies. Statistical analysis was conducted using one-way ANOVA to determine the significance of differences between experimental groups, ensuring the robustness of the results. The IC50 values for cytotoxicity were calculated using GraphPad Prism software, allowing for accurate comparisons between treatments.
Novel Aspects
The study introduced a novel pH-responsive drug delivery system that utilized BSA as a carrier for both hydrophilic and hydrophobic drugs, enhancing their solubility and therapeutic efficacy. The encapsulation efficiency was significantly higher compared to traditional nano-delivery systems, with 77% for Dox and 18% for BeA. The dual-drug system exhibited a synergistic cytotoxic effect on NSCLC cells, which was quantitatively confirmed through CI analysis. Moreover, the system demonstrated a targeted release profile, with faster drug release observed in acidic conditions (pH 6.8), characteristic of the tumor microenvironment. This approach not only improves the therapeutic index of existing drugs but also has the potential to mitigate the development of chemoresistance, offering a promising alternative to current treatment strategies.
By utilizing this methodology, the research aimed to address the limitations of traditional chemotherapy by providing a more effective and less toxic treatment option for lung cancer patients, thereby improving overall therapeutic outcomes.
Conclusion
The successful development of the BSA-(BeA+Dox) drug delivery system was achieved through a combination of innovative formulation techniques and careful characterization of the nanoparticles. By utilizing the oil-in-water micro-emulsion method, researchers were able to effectively encapsulate both doxorubicin (Dox) and betulinic acid (BeA) within bovine serum albumin (BSA) nanoparticles. This approach not only enhanced the solubility and stability of the drugs but also facilitated targeted delivery to non-small cell lung cancer (NSCLC) cells, addressing significant challenges faced by traditional chemotherapy methods.
Key highlights of this study include the demonstration of high encapsulation efficiencies—77% for Dox and 18% for BeA—along with a synergistic cytotoxic effect observed in A549 NSCLC cells. The in vitro drug release profiles indicated a pH-responsive behavior, with faster drug release in acidic conditions, mimicking the tumor microenvironment. Additionally, the study revealed that the BSA-(BeA+Dox) system significantly reduced the expression of chemoresistance markers such as the epidermal growth factor receptor (EGFR), suggesting a potential means to overcome drug resistance.
Overall, the findings underscore the potential of this innovative drug delivery system to enhance the therapeutic efficacy of existing anticancer agents while minimizing their side effects. Continued exploration of this approach may pave the way for more effective and less toxic treatment options for patients battling lung cancer.
Reference:
Torres-Martinez, Zally, et al. “A Synergistic pH-Responsive serum albumin-based drug delivery system loaded with doxorubicin and pentacyclic triterpene betulinic acid for potential treatment of NSCLC.” BioTech 12.1 (2023): 13.
