Editor: Tiffany
A new nasal delivery system using solid lipid nanoparticles significantly improves the brain targeting of atomoxetine, offering a potential advancement in the treatment of ADHD by overcoming limitations of oral administration.
Highlights
- Research Question:
Can a nasal delivery system using solid lipid nanoparticles improve the brain targeting and bioavailability of atomoxetine for treating ADHD? - Research Difficulties:
Atomoxetine’s oral bioavailability is limited by first-pass metabolism, and its effectiveness is hindered by the blood-brain barrier, necessitating an alternative delivery route. - Key Findings:
The optimized nasal inserts achieved brain targeting efficiencies of up to 211.3% and drug transport percentages of 52.7%, significantly outperforming oral and intraperitoneal administration. - Innovative Aspects:
The study introduced lyophilized nasal inserts containing atomoxetine-loaded solid lipid nanoparticles, utilizing Compritol-based SLNs and HPMC-based inserts for enhanced mucoadhesion and drug release. - Importance of the Study:
This research offers a promising alternative to oral atomoxetine delivery, potentially improving ADHD treatment outcomes by increasing drug availability in the brain and bypassing metabolic barriers.
Challenges in ADHD Treatment with Atomoxetine
Attention-Deficit/Hyperactivity Disorder (ADHD) is a common neurodevelopmental disorder affecting millions of children and adults worldwide. It is defined by persistent symptoms including inattention, hyperactivity, and impulsivity. These symptoms can impair an individual’s ability to focus, lead to impulsive actions, or result in activity levels that disrupt daily functioning. Treatment options for ADHD include stimulant and non-stimulant medications, with atomoxetine (ATM) serving as a widely used non-stimulant. Atomoxetine acts as a selective norepinephrine reuptake inhibitor, modulating neurotransmitter levels in the brain to reduce ADHD symptoms.
However, atomoxetine’s effectiveness is limited by delivery challenges. Although it is highly water-soluble and demonstrates good intestinal permeability, its oral bioavailability is restricted to 63% in extensive metabolizers and 94% in poor metabolizers due to first-pass metabolism in the liver. This process decreases the amount of active drug entering systemic circulation. Additionally, atomoxetine must cross the blood-brain barrier (BBB)—a selective barrier that limits the entry of many substances into the brain—to treat ADHD effectively. These factors underscore the need for alternative delivery methods to improve the drug’s performance.
Developing a Nasal Delivery System for Enhanced Brain Targeting
A study published in 2023 in the journal Pharmaceuticals investigated a nasal delivery system designed to address these limitations. Conducted by Mahmoud H. Teaima, Merhan Taha El-Nadi, Raghda Rabe Hamed, Mohamed A. El-Nabarawi, and Rehab Abdelmonem from Cairo University, the Egyptian Drug Authority, and Misr University for Science and Technology, the research aimed to enhance atomoxetine’s bioavailability and brain targeting efficiency. The team developed lyophilized nasal inserts containing atomoxetine-loaded solid lipid nanoparticles (ATM-SLNs) to bypass first-pass metabolism and the blood-brain barrier, delivering the drug directly to the brain through the nasal route.
Formulation and Evaluation of Atomoxetine-Loaded Nasal Inserts
The researchers followed a structured experimental approach to develop and evaluate the nasal delivery system. The methodology consisted of the following steps:
- Preparation of ATM-SLNs: Atomoxetine-loaded solid lipid nanoparticles were created using lipids (e.g., Compritol 888 ATO, stearic acid) and surfactants (e.g., Span 60, Pluronic F127). A hot melt emulsification technique, followed by stirring and ultrasonication, was used to produce stable nanoparticles.
- Characterization of ATM-SLNs: The nanoparticles were analyzed for entrapment efficiency (EE%), particle size (PS), zeta potential (ZP), and in-vitro drug release (Q30min) to assess their suitability for delivery.
- Optimization: A full factorial design tested variables including lipid type, lipid-to-drug ratio, surfactant ratio, and sonication time. Two formulations, F7 and F9, were identified as optimal based on their performance.
- Preparation of Nasal Inserts: The selected ATM-SLNs (F7 and F9) were incorporated into lyophilized nasal inserts using mucoadhesive polymers such as hydroxypropyl methylcellulose (HPMC K100m) or NOVEON AA1, with glycine as a collapse-protecting agent.
- Evaluation of Nasal Inserts: The inserts were examined for drug content, in-vitro release, mucoadhesion strength, disintegration time, and residual water content to confirm their effectiveness.
- In-Vivo Studies: An in-vivo pharmacokinetics study in rats compared the brain targeting efficiency of the nasal inserts to oral and intraperitoneal administration routes.
Key Experiments and Findings
1. Optimization of ATM-SLNs Formulations
- Procedure: Sixteen ATM-SLN formulations were prepared, varying lipid types, surfactant ratios, and sonication times, and evaluated for EE%, PS, ZP, and Q30min.
- Result: Formulations F7 and F9 performed best. F7 had an EE% of 83.06%, PS of 795.60 nm, and ZP of -28.39 mV, while F9 showed an EE% of 55.20%, PS of 392.10 nm, and ZP of -16.5 mV.
- Observation: Using Compritol 888 ATO and a higher Pluronic F127 to Span 60 ratio improved entrapment efficiency and nanoparticle stability.
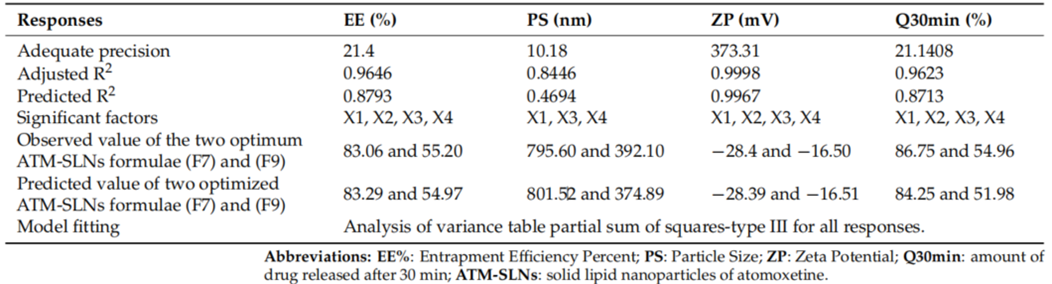
Table 1. Experimental runs, independent variables, and measured responses of the 24 full factorial experimental design of ATM-loaded SLNs.
2. Characterization of Lyophilized Nasal Inserts
- Procedure: Eight nasal insert formulations were developed from F7 and F9 SLNs, with variations in polymer type (HPMC K100m or NOVEON AA1) and glycine concentration, and tested for multiple parameters.
- Result: Inserts S4 and S8, made with HPMC K100m and higher glycine levels, showed drug content near 100%, disintegration times of 20-30 seconds, and mucoadhesion strengths of 8436.6 dyne/cm² (S4) and 9025.2 dyne/cm² (S8).
- Observation: HPMC-based inserts provided better drug release and mucoadhesion than NOVEON AA1-based inserts, particularly with increased glycine.
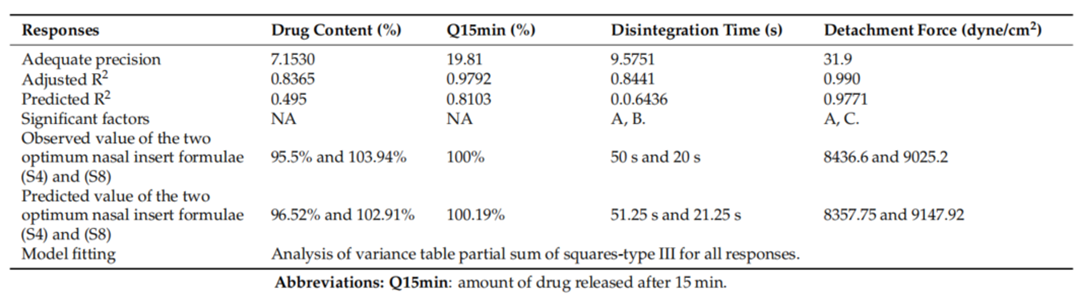
Table 2. Experimental runs, independent variables, and measured responses of the 23 full factorial experimental design of lyophilized nasal insert of ATM-loaded SLNs.
3. In-Vivo Pharmacokinetics Study
- Procedure: Nasal inserts S4 and S8 were tested in rats alongside an oral solution, intraperitoneal injection, and nasal insert with pure atomoxetine. Drug concentrations in plasma and brain tissue were measured.
- Result: Nasal inserts S8 and S4 achieved brain targeting efficiencies of 211.3% and 177.42%, respectively, with drug transport percentages of 52.7% and 43.64%, outperforming other methods.
- Observation: The nasal delivery system enhanced atomoxetine’s brain delivery, bypassing first-pass metabolism and the blood-brain barrier.

Table 3. Pharmacokinetic parameters of atomoxetine in plasma and brain homogenate tissues following its intranasal (IN) route (lyophilized inserts of ATM-loaded SLNs (S4 and S8) and lyophilized insert of pure atomoxetine), intravenous (IV) route and oral route (Atomorelax® oral solution).
Implications for ADHD Therapy and Future Directions
This study developed a nasal delivery system for atomoxetine using solid lipid nanoparticles integrated into lyophilized nasal inserts. The system, optimized with Compritol-based SLNs and HPMC-based inserts, improved bioavailability and brain targeting compared to oral administration. Published in 2023, the findings suggest a promising alternative for ADHD treatment by increasing the drug’s delivery to the brain, addressing limitations posed by first-pass metabolism and the blood-brain barrier. This approach may enhance the management of ADHD symptoms through more efficient drug delivery.
Reference:
Teaima, Mahmoud H., et al. “Lyophilized nasal inserts of atomoxetine HCl solid lipid nanoparticles for brain targeting as a treatment of attention-deficit/hyperactivity disorder (ADHD): a pharmacokinetics study on rats.” Pharmaceuticals 16.2 (2023): 326.
