Editor: Tiffany
A new nanoconjugate combining hyaluronic acid and graphene oxide quantum dots has been developed to improve metformin’s delivery and effectiveness in treating inflammation associated with type 2 diabetes and obesity, offering a potential dual-action therapy.
Highlights
- Research Question:
Can a nanoconjugate of hyaluronic acid and graphene oxide quantum dots improve the targeted delivery and efficacy of metformin in treating meta-inflammation in type 2 diabetes and obesity? - Research Difficulties:
Enhancing metformin’s bioavailability and achieving specific targeting to inflamed tissues posed significant challenges, as metformin in its standard form lacks precise delivery mechanisms and has limited anti-inflammatory effects. - Key Findings:
The nanoconjugate (GOQD-HA-Met) reduced proinflammatory cytokine expression, restored antioxidant status, and improved metabolic parameters in both cell-based and animal models, outperforming free metformin at lower doses. - Innovative Aspects:
The study introduces a dual-action nanocarrier that enhances metformin delivery while independently contributing to reduced inflammation and oxidative stress through its hyaluronic acid and graphene oxide quantum dot components. - Importance of the Study:
This approach offers a promising strategy to target meta-inflammation, a key driver of insulin resistance in type 2 diabetes and obesity, potentially leading to improved treatment outcomes.
Meta-Inflammation in Type 2 Diabetes and Obesity: A Therapeutic Challenge
Type 2 diabetes mellitus (T2DM) is a widespread metabolic disorder marked by elevated blood sugar levels due to insulin resistance, where the body’s cells do not respond effectively to insulin. Obesity significantly increases the risk of developing T2DM, and both conditions are closely tied to a chronic, low-grade inflammatory state known as meta-inflammation. This inflammation predominantly affects adipose tissue, leading to adipose tissue inflammation (ATI), and the liver, causing non-alcoholic steatohepatitis (NASH). In these conditions, immune cells such as macrophages become activated and release proinflammatory cytokines, including interleukin-1 beta (IL-1β), tumor necrosis factor-alpha (TNF-α), and interleukin-6 (IL-6). These cytokines impair insulin signaling pathways, worsening insulin resistance and creating a self-perpetuating cycle of metabolic dysfunction.
Adipose tissue inflammation arises from the accumulation of excess fat, which triggers macrophage infiltration and the subsequent release of inflammatory mediators. Similarly, NASH involves liver inflammation driven by fat accumulation, further contributing to systemic insulin resistance. Together, ATI and NASH amplify the inflammatory burden in T2DM and obesity, making them critical therapeutic targets.
The primary treatment for T2DM is metformin, a drug that reduces blood glucose levels and has some anti-inflammatory effects. Despite its widespread use, metformin faces significant challenges. Its bioavailability ranges from 50-60%, meaning only about half of the administered dose reaches the bloodstream, necessitating high daily doses—often up to 2000 mg—to achieve therapeutic effects. Moreover, metformin does not specifically target inflamed tissues like adipose tissue or the liver, limiting its ability to address the underlying meta-inflammation driving insulin resistance. These limitations highlight the need for innovative approaches to enhance metformin’s delivery and efficacy in treating T2DM and obesity.
Developing a Targeted Nanoconjugate for Enhanced Metformin Delivery
The challenges of targeting inflammation in T2DM and obesity with current treatments stem from their lack of specificity and poor tissue penetration. Metformin, while effective at managing blood sugar, does not concentrate in inflamed areas where meta-inflammation occurs, reducing its impact on ATI and NASH. To overcome this, researchers aimed to develop a novel drug delivery system that directs metformin to these specific sites of inflammation.
The study’s primary objective was to create a CD44-targeted nanocomposite using hyaluronic acid-functionalized graphene oxide quantum dots (GOQD-HA) to deliver metformin precisely to inflamed tissues. The CD44 receptor is overexpressed on macrophages in inflamed adipose tissue and liver, making it an ideal target. By functionalizing graphene oxide quantum dots (GOQD) with hyaluronic acid (HA)—a natural ligand for CD44—and loading metformin onto this structure (GOQD-HA-Met), the researchers sought to improve the drug’s bioavailability and anti-inflammatory effects at lower doses. This targeted approach was designed to enhance metformin’s therapeutic potential by concentrating it where inflammation is most active.
The research was conducted by a team including Kunal Sarkar, Sarbashri Bank, Arindam Chatterjee, Koushik Dutta, Anwesha Das, Santanu Chakraborty, Nirvika Paul, Jit Sarkar, Sriparna De, Sudakshina Ghosh, Krishnendu Acharyya, Dipankar Chattopadhyay, and Madhusudan Das, primarily affiliated with the University of Calcutta. Their findings were published in the Journal of Nanobiotechnology in 2023, marking a significant advancement in nanomedicine for metabolic disorders.
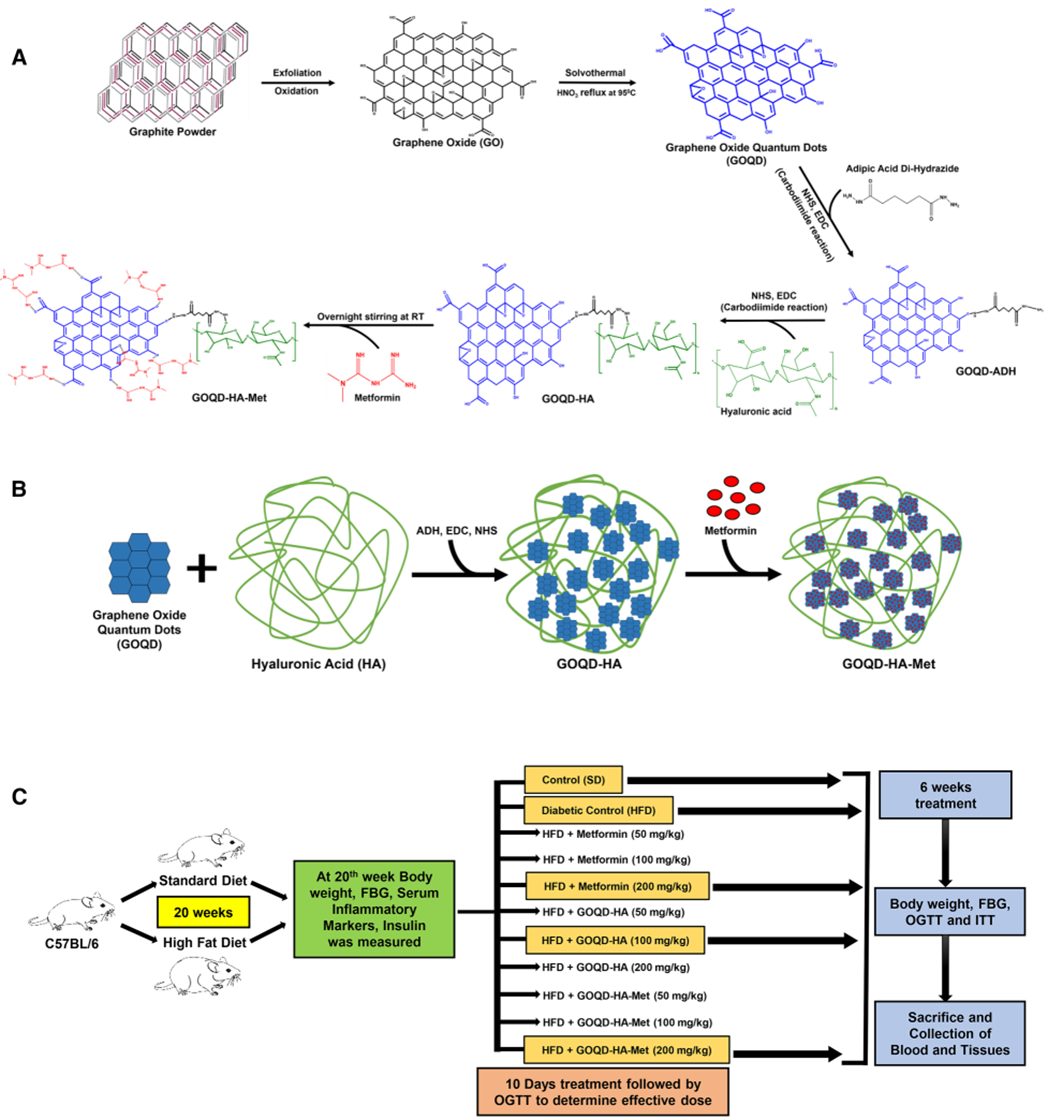
Figure 1. (A) Synthesis scheme of Metformin loaded Graphene Oxide Quantum dots- Hyaluronic acid nanocomposite (GOQD-HA-Met). (B) Simplified synthesis scheme of GOQD-HA-Met. (C) Schematic representation of animal study design.
Experimental Validation of the Nanoconjugate’s Efficacy
The study employed a structured experimental process to synthesize and test the GOQD-HA-Met nanocomposite, using both in vitro and in vivo approaches to evaluate its efficacy.
Experimental Process
- Synthesis of GOQD: Graphene oxide was produced from graphite using a modified Hummers’ method, followed by acid treatment and dialysis to form graphene oxide quantum dots (GOQD).
- Conjugation with HA: GOQD was functionalized with hyaluronic acid via a carbamide reaction, creating GOQD-HA, which targets the CD44 receptor.
- Loading Metformin: Metformin was adsorbed onto GOQD-HA through mixing and centrifugation, forming the final nanocomposite, GOQD-HA-Met.
- In Vitro Studies: RAW264.7 macrophage cells were exposed to palmitic acid (250 μM) to induce inflammation, then treated with GOQD-HA-Met (130 μg/ml, equivalent to 0.5 mM metformin) for 8 hours. Inflammatory markers were measured using real-time polymerase chain reaction (RT-PCR) and immunofluorescence.
- In Vivo Studies: Diet-induced obese (DIO) mice were orally administered GOQD-HA-Met (200 mg/kg, equivalent to 100 mg/kg metformin) daily for 6 weeks. Blood and tissue samples were analyzed for inflammatory cytokines, antioxidant enzyme activity (catalase [CAT] and superoxide dismutase [SOD]), and metabolic parameters like glucose tolerance and insulin sensitivity.
Key Experiments and Findings
1. In Vitro Anti-Inflammatory Effects
- Procedure: RAW264.7 cells were treated with palmitic acid to simulate inflammation, followed by GOQD-HA-Met or free metformin (1 mM) for comparison. Levels of IL-1β, TNF-α, and IL-6 were quantified.
- Result: GOQD-HA-Met reduced the expression of IL-1β, TNF-α, and IL-6 more effectively than free metformin, despite using a lower equivalent dose of the drug (0.5 mM vs. 1 mM).
- New Finding: The targeted delivery via GOQD-HA enhanced metformin’s anti-inflammatory effects, suggesting that the nanocomposite improves drug potency by concentrating it at sites of inflammation.
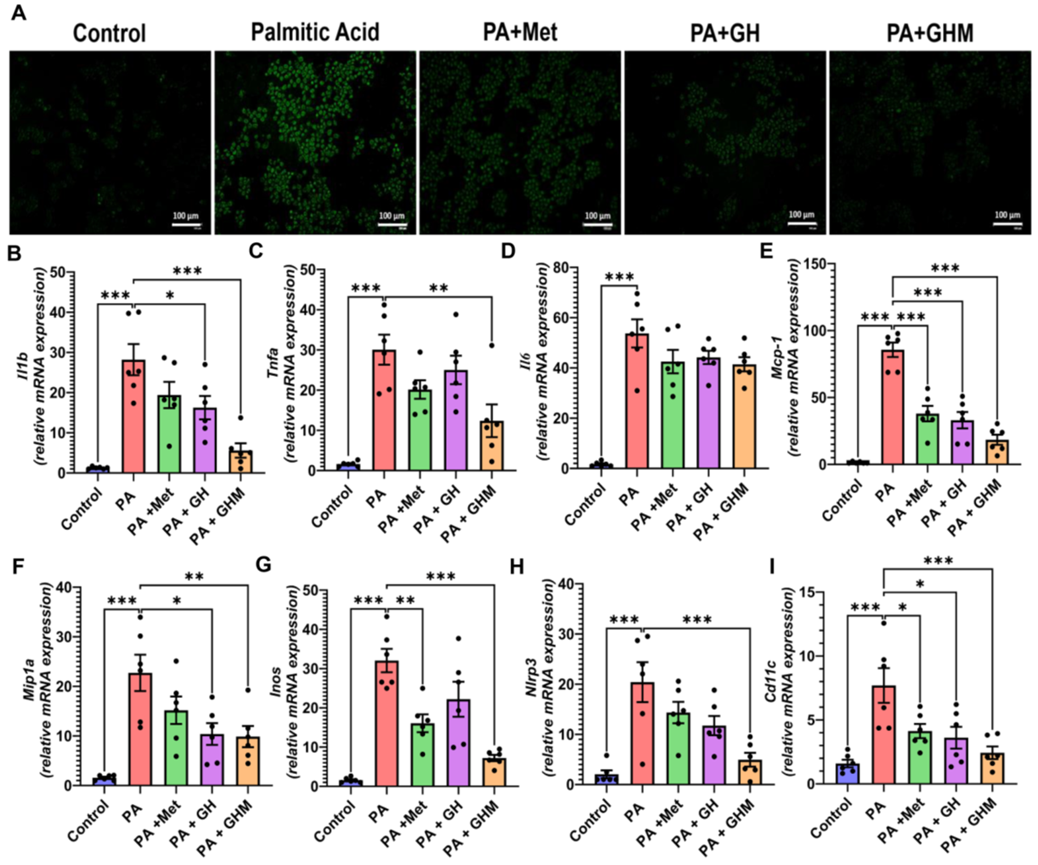
Figure 2. In vitro anti inflammatory effects of GOQD HA Met.
2. In Vivo Efficacy in DIO Mice
- Procedure: DIO mice received GOQD-HA-Met for 6 weeks, after which plasma levels of inflammatory cytokines, antioxidant enzyme activity, and metabolic markers were assessed.
- Result: Treatment decreased plasma IL-1β, TNF-α, and IL-6 levels, increased CAT and SOD activity, and improved glucose tolerance and insulin sensitivity compared to untreated controls or free metformin-treated mice.
- New Finding: GOQD-HA-Met not only delivered metformin effectively but also exhibited inherent anti-inflammatory and antioxidant properties, enhancing overall therapeutic outcomes beyond metformin alone.
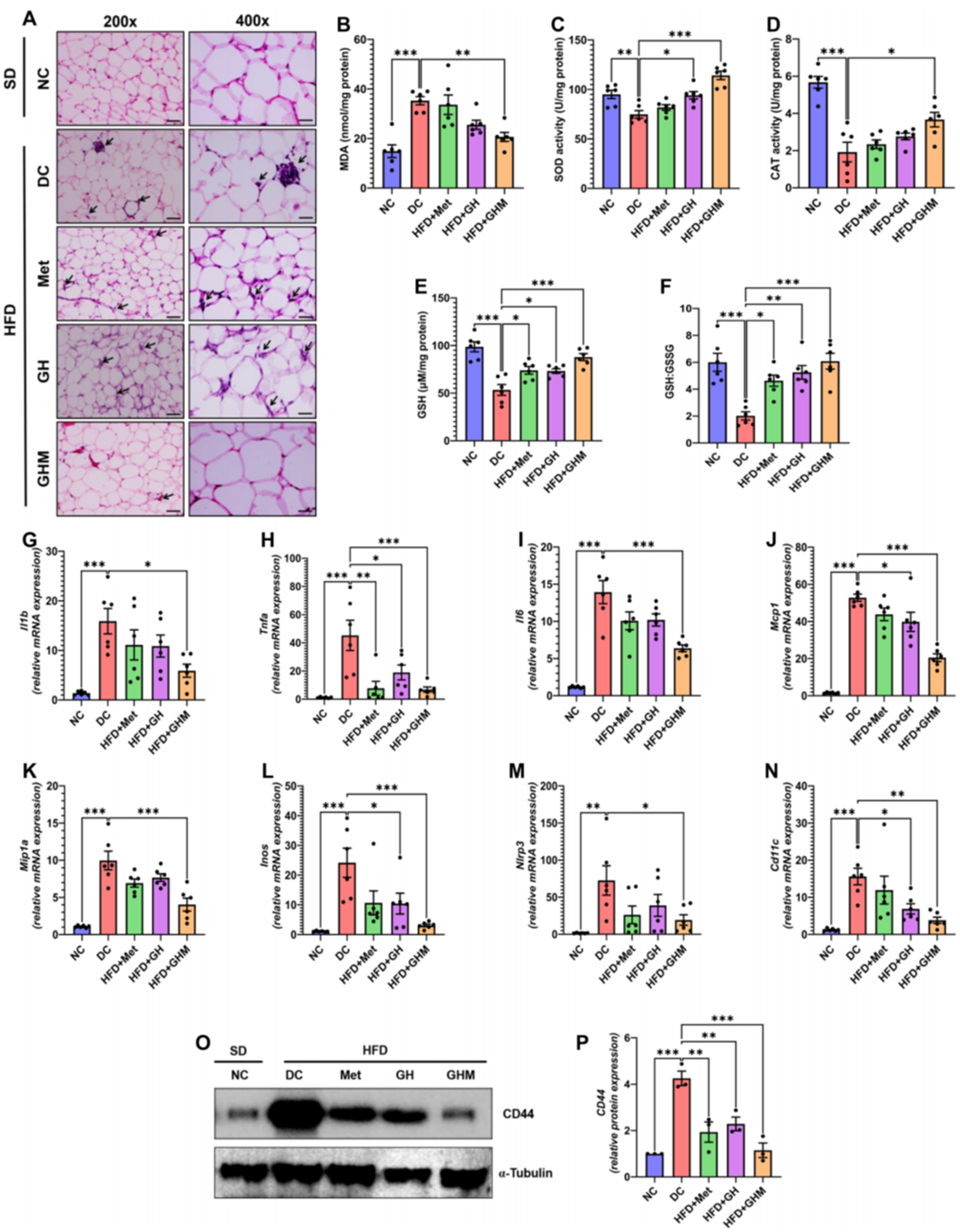
Figure 3. GOQD-HA-Met efciently attenuated HFD induced adipose tissue infammation.
These results demonstrate that the nanocomposite successfully targets inflamed tissues, amplifying metformin’s effects while addressing oxidative stress, a key component of meta-inflammation.
Therapeutic Potential of the Nanoconjugate in Treating Meta-Inflammation
The study achieved a significant milestone by developing GOQD-HA-Met, a CD44-targeted nanocomposite that delivers metformin directly to inflamed tissues in T2DM and obesity. In vitro tests with RAW264.7 cells showed that GOQD-HA-Met reduced inflammatory cytokine expression more effectively than free metformin at a lower dose. In vivo experiments with DIO mice confirmed these findings, demonstrating reductions in inflammation, enhancements in antioxidant defenses, and improvements in metabolic health after 6 weeks of treatment.
A standout feature of this nanocomposite is its dual-action capability. Beyond serving as a delivery vehicle, GOQD-HA itself contributes anti-inflammatory and antioxidant effects. The graphene oxide quantum dots scavenge reactive oxygen species, while hyaluronic acid may modulate inflammation, synergizing with metformin to produce a more robust therapeutic response. This dual mechanism enhances metformin’s efficacy, overcoming its limitations of poor bioavailability and lack of specificity.
The significance of this research lies in its novel insight: the nanocarrier not only transports metformin but also amplifies its effectiveness, offering a more efficient treatment strategy for meta-inflammation. This approach has broader implications, suggesting potential applications for other inflammatory diseases where targeted drug delivery could improve outcomes. By addressing the root causes of insulin resistance in T2DM and obesity, GOQD-HA-Met represents a promising advancement in nanomedicine, paving the way for future therapies that combine drug delivery with inherent therapeutic properties.
Reference:
Sarkar, Kunal, et al. “Hyaluronic acid-graphene oxide quantum dots nanoconjugate as dual purpose drug delivery and therapeutic agent in meta-inflammation.” Journal of Nanobiotechnology 21.1 (2023): 246.
