Editor: Tiffany
Researchers at Johns Hopkins University have developed a novel gene therapy approach using biodegradable nanoparticles delivered directly to liver tumors via the hepatic artery, achieving significantly higher tumor targeting and gene expression compared to traditional intravenous methods.
Highlights
- Research Question:
Can biodegradable nanoparticles be used to deliver gene therapy specifically to hepatocellular carcinoma (HCC) tumors while minimizing effects on healthy liver tissue? - Research Difficulties:
Systemic delivery methods for gene therapy often result in poor tumor targeting and potential toxicity to healthy tissues, particularly in the liver, which has a unique dual blood supply. - Key Findings:
Intra-arterial injection of nanoparticles via the hepatic artery led to a 27.9-fold increase in gene expression within HCC tumors compared to a negligible 1.61-fold increase with intravenous delivery. - Innovative Aspects:
This study is the first to demonstrate the use of polymeric nanoparticles delivered through the hepatic artery for targeted gene therapy in an orthotopic rat model of HCC. - Importance of the Study:
The research offers a promising new strategy for HCC treatment that could enhance therapeutic efficacy and reduce systemic side effects, addressing critical limitations of current therapies like TACE.
Challenges in Treating Liver Cancer: The Need for Targeted Therapies
Hepatocellular carcinoma (HCC) is recognized as a leading cause of cancer-related deaths worldwide, presenting a significant global health challenge. This form of liver cancer is notoriously difficult to manage, largely due to the liver’s unique physiological characteristics, which complicate treatment efforts. While the specific symptoms of HCC are not detailed in the study, the disease’s association with liver dysfunction suggests potential manifestations such as jaundice, abdominal pain, and weight loss. However, the primary focus remains on the therapeutic hurdles posed by HCC.
Current treatment options for HCC include trans arterial chemoembolization (TACE), a locoregional therapy that delivers chemotherapy directly to the tumor through the hepatic artery, followed by an embolic agent to obstruct the tumor’s blood supply. Despite its targeted approach, TACE has limitations. Research indicates that the addition of chemotherapeutic agents to TACE provides only modest benefits, possibly due to the poor pharmacokinetics of small molecule drugs, which suffer from short half-lives and potential systemic toxicity. An alternative method, drug eluting bead TACE (DEB-TACE), has been explored, but it has not demonstrated proven advantages over conventional TACE. These shortcomings highlight the urgent need for innovative, more effective strategies to combat HCC.
Developing a Nanoparticle-Based Gene Delivery System for HCC
A team of researchers at Johns Hopkins University School of Medicine, led by Hannah J Vaughan and Jordan J Green, sought to address these challenges by developing a novel gene delivery system using biodegradable nanoparticles for HCC treatment. Their primary aim was to create a targeted therapy capable of delivering genetic material directly to HCC tumors while sparing healthy liver tissue. The study, published in the International Journal of Nanomedicine in 2023, was driven by the need to overcome the limitations of systemic delivery methods, which often result in poor tumor targeting and unwanted side effects.
To achieve this, the researchers proposed using intra-arterial injection, a method that leverages the hepatic artery to directly access the tumor’s blood supply. This approach was intended to enhance the accumulation of therapeutic agents within the tumor and reduce systemic toxicity. The objectives centered on testing the efficacy of this nanoparticle-based gene delivery system in a rat model of HCC, with the goal of improving treatment precision and outcomes.
Experimental Approach: From Polymer Synthesis to In Vivo Testing
The study was conducted in several distinct phases, each designed to evaluate the potential of the nanoparticle system:
- Polymer Synthesis and Nanoparticle Formation: The researchers synthesized three types of poly(beta-amino ester) (PBAE) polymers—labeled 446, 457, and 536—and selected PBAE 536 as the optimal polymer for nanoparticle formation based on its suitability for gene delivery.
- In Vitro Testing: Rat HCC cells (N1-S1) were treated with nanoparticles carrying green fluorescent protein (GFP) DNA. Various doses and weight ratios of the three polymer formulations were tested to assess transfection efficiency and cell viability.
- Nanoparticle Characterization: The physical properties of the nanoparticles, including size and surface charge, were analyzed to confirm their stability and appropriateness for in vivo application.
- In Vivo Studies: Rats with orthotopic N1-S1 HCC tumors received nanoparticle injections via either the intra-arterial or intravenous route. The intra-arterial method involved surgical access to the hepatic artery, mirroring the clinical TACE procedure.
Two key experiments provided critical insights:
- In Vitro Transfection
- Procedure: N1-S1 HCC cells were exposed to nanoparticles formulated with PBAE polymers 446, 457, and 536, each carrying GFP DNA, across different doses and weight ratios.
- Result: Over 50% of the cells expressed GFP with cell viability exceeding 70% for most formulations. However, the highest doses and weight ratios of polymers 457 and 536 showed reduced viability.
- Finding: PBAE 536 was identified as an effective polymer for HCC cell transfection, offering a balance of intermediate transfection efficacy and good cell viability.
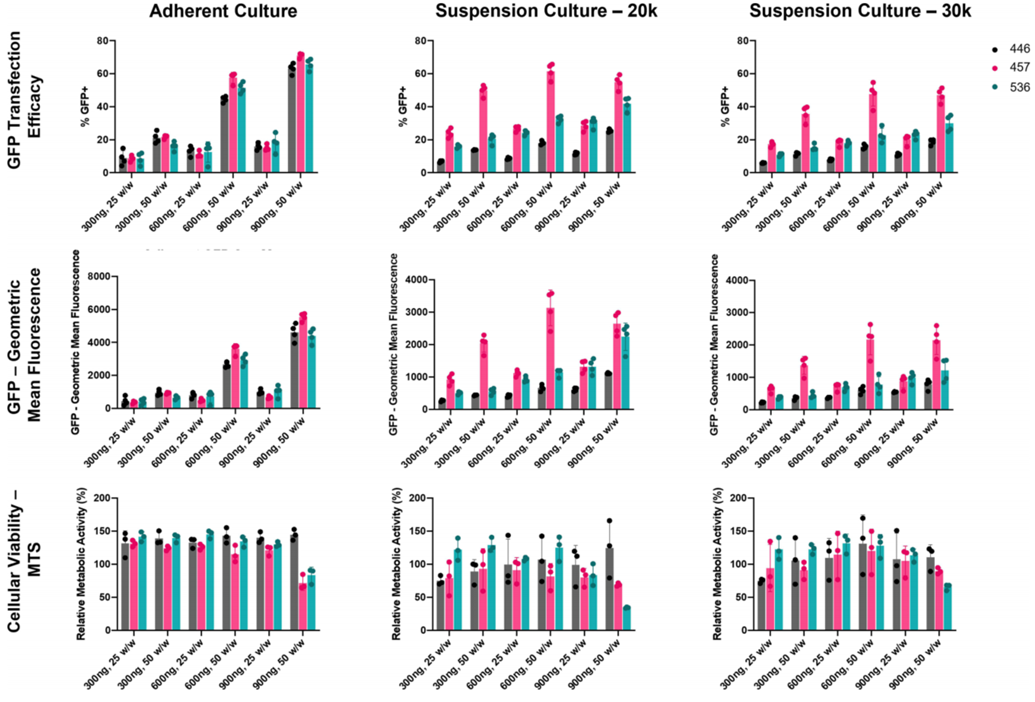
Figure 1. PBAE NP Screening in N1-S1 cells.
- In Vivo Biodistribution and Transfection
- Procedure: Nanoparticles carrying the firefly luciferase gene were administered to rats with HCC tumors via either intra-arterial injection (through the hepatic artery) or intravenous injection.
- Result: Intra-arterial delivery resulted in higher nanoparticle accumulation in both the tumor and surrounding liver parenchyma, achieving a 27.9-fold increase in average radiance (indicating successful gene expression) within the tumor. In contrast, intravenous delivery showed accumulation primarily in healthy liver tissue, with only a 1.61-fold increase in radiance, indicating negligible transfection in the tumor.
- Finding: Intra-arterial injection significantly enhanced both biodistribution and transfection efficacy compared to intravenous delivery, marking a notable advancement in targeted gene therapy for HCC.
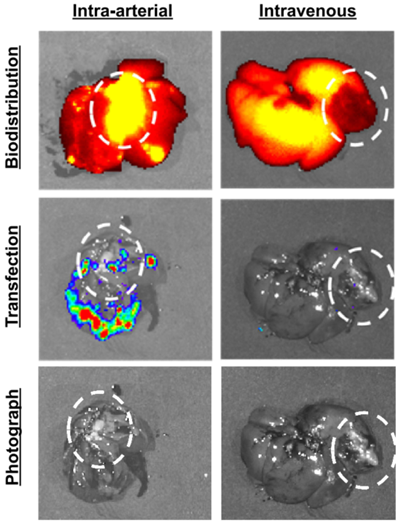
Figure 2. NIR-fLuc NP biodistribution and transfection following intra-arterial or intravenous injection in animals with N1-S1 tumors (circled).
Advancing HCC Treatment: Implications of Targeted Nanoparticle Delivery
This research presents a promising new strategy for HCC treatment by utilizing biodegradable nanoparticles to deliver gene therapy directly to liver tumors via intra-arterial injection. The study demonstrated that this method achieves superior targeting and gene expression within the tumor compared to traditional intravenous delivery, with a 27.9-fold increase in radiance versus a 1.61-fold increase, respectively. By leveraging the hepatic artery—the tumor’s primary blood supply—the approach enhances the accumulation of therapeutic agents in the tumor while reducing exposure to healthy tissues, potentially lowering systemic side effects.
The significance of this work lies in its pioneering use of polymeric nanoparticles delivered through the hepatic artery in an orthotopic rat model of HCC, a first in the field of nanomedicine. This innovation addresses key limitations of existing therapies like TACE and offers a pathway toward more precise and effective treatments. The findings suggest that this nanoparticle-based system could improve HCC treatment efficacy and safety, laying the groundwork for future clinical investigations. As a significant advancement in liver cancer therapy, this research provides hope for better management of a disease that continues to claim numerous lives globally.
Reference:
Vaughan, Hannah J., et al. “Non-viral gene delivery to hepatocellular carcinoma via intra-arterial injection.” International journal of nanomedicine (2023): 2525-2537.
