Editor: Nina
The study demonstrates that lactoferrin-coated biosynthesized selenium nanoparticles (ALF-Se NPs) effectively target and induce apoptosis in various human cancer cell lines while minimizing toxicity to normal cells, suggesting a promising avenue for the development of selective cancer therapies.
Key Preview
Research Question
The study investigates the effectiveness of lactoferrin-coated biosynthesized selenium nanoparticles (ALF-Se NPs) in selectively targeting and inducing apoptosis in various human cancer cell lines while minimizing toxicity to normal cells.
Research Design and Strategy
The researchers employed a combination of bioengineering techniques to create selenium nanoparticles using the yeast Rhodotorula sp. and subsequently modified them by coating with bovine lactoferrin. This design aims to enhance the anticancer properties of selenium nanoparticles and improve their selectivity towards cancer cells.
Method
The study utilized a series of in vitro assays involving cancer cell lines (MCF-7, HepG-2, and Caco-2) to assess the cytotoxic effects of ALF-Se NPs compared to free lactoferrin and selenium nanoparticles. Morphological changes, apoptosis rates, and gene expression profiles were analyzed to elucidate the mechanism of action.
Key Results
ALF-Se NPs demonstrated significantly higher anticancer activity, with an IC50 value of approximately 63.10 μg/mL across tested cancer cell lines, showing over 64 times selectivity against cancer cells compared to normal cells. The treatment led to upregulation of pro-apoptotic genes (p53) and downregulation of anti-apoptotic genes (Bcl-2, MMP-9, and VEGF).
Significance of the Research
This research provides a promising avenue for the development of targeted cancer therapies using biocompatible nanoparticles, highlighting the potential of combining nanotechnology with biomedicine to enhance treatment efficacy and reduce side effects.
Introduction
The quest for effective cancer therapies has evolved significantly over the years, with a growing focus on targeted treatment strategies that minimize harm to normal cells. Traditional chemotherapy often results in severe side effects due to its lack of selectivity. Recent advancements in nanotechnology have opened new frontiers in cancer treatment, allowing for the development of drug delivery systems that can selectively target tumor tissues.
The current study introduces a novel approach combining selenium nanoparticles with lactoferrin, a bioactive protein known for its immunomodulatory effects. This research aims to address the critical challenge of achieving selective cytotoxicity against cancer cells while sparing healthy tissues. By leveraging the unique properties of lactoferrin and selenium nanoparticles, the researchers aimed to enhance the anticancer efficacy of this nanocombination.
Research Team and Objective
The study was conducted by a collaborative research team comprising Esmail M. El-Fakharany, Marwa M. Abu-Serie, Amany Ibrahim, and Marwa Eltarahony from the Genetic Engineering and Biotechnology Research Institute (GEBRI) and Ain Shams University in Egypt. Published in the journal Scientific Reports, the paper titled “Anticancer activity of lactoferrin-coated biosynthesized selenium nanoparticles for combating different human cancer cells via mediating apoptotic effects” aims to investigate the anticancer potential of ALF-Se NPs and elucidate their mechanism of action.
The objective of the research is to develop a highly selective anticancer agent that minimizes collateral damage to normal cells while efficiently targeting various human cancer cell lines.
Experimental Process
Biosynthesis of Selenium Nanoparticles (Se NPs)
- Key Steps: The study employed the yeast strain Rhodotorula sp. MZ312359 to bio-reduce sodium selenite (Na2SeO3) into selenium nanoparticles. This was achieved by inoculating the yeast in a liquid medium and adding the selenium precursor. The visual change from yellow to red in the reaction mixture indicated the successful reduction process.
- Results and Key Data: The biosynthesis was confirmed optically, with a maximum absorption peak observed at 395 nm in the UV-Vis spectrum, indicating the formation of Se NPs. The size of the nanoparticles, assessed by Transmission Electron Microscopy (TEM), ranged from 18-40 nm.
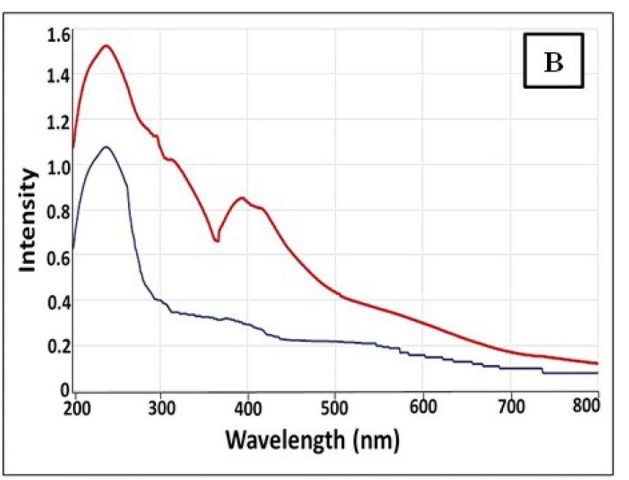
Figure 1. UV–Vis spectroscopy of microbially synthesized Se NPs by strain Rhodotorula sp. MNR.
- Significance of the Result: The successful biosynthesis of Se NPs using a biological method provides an eco-friendly alternative to chemical synthesis, harnessing the capabilities of microorganisms to produce nanoparticles with potential applications in biomedicine.
- Key Innovations: This method highlights the use of a specific yeast strain for the biosynthesis of selenium nanoparticles, which differs from traditional chemical synthesis methods, thus reducing toxicity and improving sustainability.
Characterization of Se NPs
- Key Steps: The synthesized Se NPs were characterized using several techniques: Scanning Electron Microscopy (SEM) and Transmission Electron Microscopy (TEM) were utilized to visualize the morphology and size distribution, while X-Ray Diffraction (XRD) analyzed the crystallinity. Fourier-Transform Infrared Spectroscopy (FTIR) was employed to investigate functional groups present on the Se NPs.
- Results and Key Data: Characterization showed the Se NPs had a spherical morphology with an average size of 29 ± 2.3 nm, and XRD indicated an amorphous structure with a major orientation at 2θ = 29.28°. FTIR results revealed functional groups such as –OH, –NH2, and –CO, indicating the presence of biomolecules that stabilize the nanoparticles.
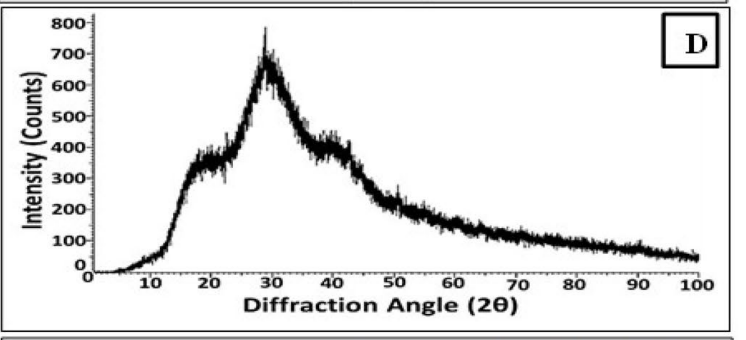
Figure 2. XRD pattern
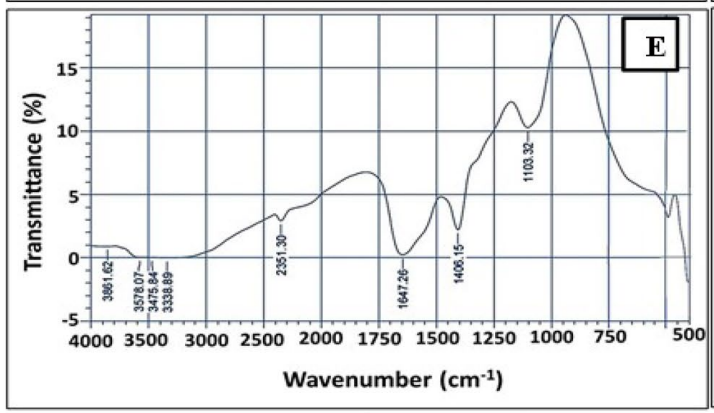
Figure 3. FTIR analysis of Se NPs
- Significance of the Result: Characterization confirms the physical and chemical properties of the synthesized nanoparticles, which are crucial for understanding their potential interactions in biological systems and therapeutic applications.
- Key Innovations: The combination of biosynthesis with advanced characterization techniques provides a comprehensive understanding of the nanoparticles, enabling insights into their stability and reactivity, which are essential for drug delivery applications.
Preparation of Lactoferrin-Coated NPs
- Key Steps: The researchers purified bovine lactoferrin (bLF) from whey using cation exchange chromatography followed by size exclusion chromatography to obtain apolactoferrin (ALF). The purified ALF was then combined with Se NPs to form ALF-Se NPs through a stirring process.
- Results and Key Data: The purity of bLF was confirmed via SDS-PAGE, showing a single band at approximately 80 kDa. The resulting ALF-Se NPs exhibited an average size of less than 200 nm and were characterized by SEM and TEM.
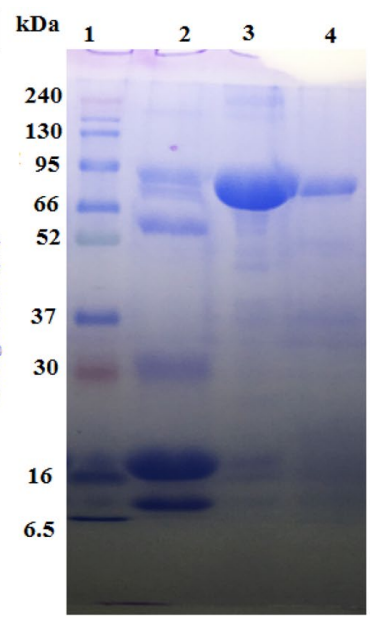
Figure 4. 12% SDSPAGE for bLF during purifcation steps. Lane 1 is protein marker, lane 2 is the raw bovine whey, lane 3 is bLF eluted from CM Sephadex C50 column and lane 4 is purifed bLF eluted from Sephacryl S200 column.
- Significance of the Result: The successful coating of Se NPs with ALF enhances the biocompatibility and stability of the nanoparticles, establishing a platform for targeted drug delivery systems that can selectively interact with cancer cells.
- Key Innovations: This step showcases the novel approach of using bioengineered nanoparticles with a functional protein coating, which can potentially improve the therapeutic efficacy of the nanoparticles.
Assessment of Anticancer Activity
- Key Steps: The cytotoxic effects of ALF, Se NPs, and ALF-Se NPs were evaluated using MTT assays on various human cancer cell lines (MCF-7, HepG-2, Caco-2) and normal cells (Wi-38). The cell viability was measured after 72 hours of treatment with serial dilutions of the nanoparticles and proteins.
- Results and Key Data: The ALF-Se NPs demonstrated a significantly lower IC50 of approximately 63.10 μg/mL across all tested cancer cell lines, with a selectivity index greater than 64, indicating a marked preference for killing cancer cells over normal cells.
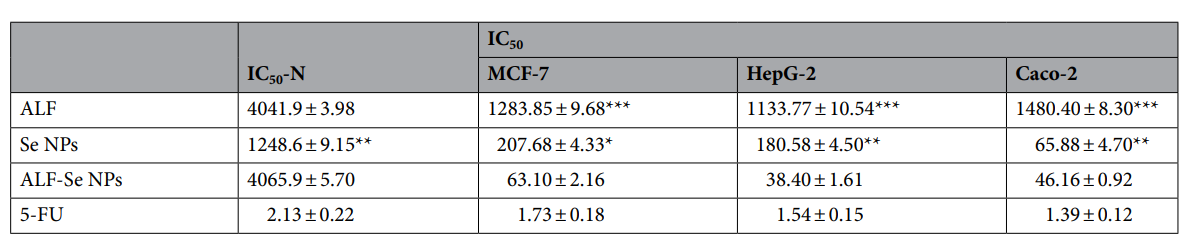
Table 1. Cytotoxicity of ALF with Se NPs in the term of IC50-N (μg/ml) and IC50 (μg/ml) against human normal cells (Wi-38) and cancer cells as compared to 5-FU. All values are expressed as mean ± SEM. ALF-Se NPs is statistically signifcant with ALF and Se NPs at p < 0.05*, p < 0.005** and p < 0.0005***.
- Significance of the Result: This result underscores the potential of ALF-Se NPs as a targeted anticancer therapy that minimizes toxicity to normal tissues, representing a significant advancement in cancer treatment strategies.
- Key Innovations: The study highlights the exceptional selectivity of the ALF-Se NPs compared to traditional chemotherapeutic agents, paving the way for more personalized and less harmful cancer treatments.
Flow Cytometric Analysis of Apoptosis
- Key Steps: Flow cytometry was utilized to quantify apoptosis in treated cancer cells using annexin V and propidium iodide (PI) staining. This allowed for the differentiation between early and late apoptotic cells.
- Results and Key Data: ALF-Se NPs induced apoptosis rates of 47.74%, 50.09%, and 52.6% in MCF-7, HepG-2, and Caco-2 cells, respectively, compared to lower rates observed with ALF and Se NPs alone.
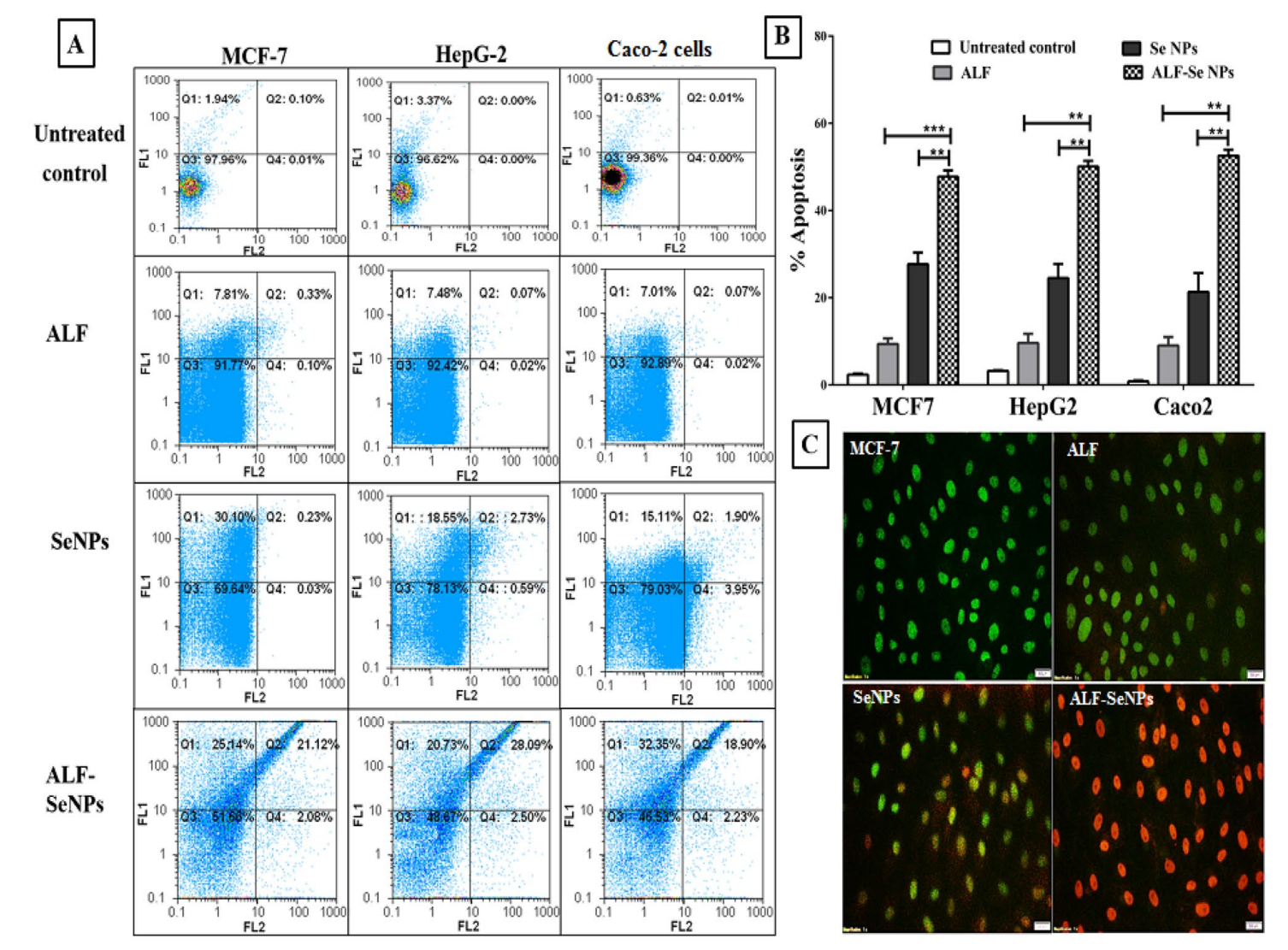
Figure 5. Apoptotic efect of ALF-Se NPs in comparison with ALF and Se NPs. (A) Flow charts of ALF, Se NPs and ALF-Se NPs-treated Caco-2, HepG-2 and MCF-7 cells relative to the untreated cells with (B) the percentage of apoptotic cell population afer staining with annexin V/propidium iodide.
- Significance of the Result: The enhanced apoptotic effect seen with ALF-Se NPs suggests a robust mechanism of action that could be leveraged for cancer therapies, enhancing the likelihood of tumor regression.
- Key Innovations: This analysis provides a detailed understanding of the mechanism by which ALF-Se NPs exert their anticancer effects, contributing to the growing body of evidence supporting the use of nanomedicine in oncology.
Gene Expression Studies
- Key Steps: The expression levels of apoptosis-related genes (p53, Bcl-2, MMP-9, VEGF) were assessed through quantitative PCR to elucidate the molecular mechanisms underlying the anticancer effects of ALF-Se NPs.
- Results and Key Data: Treatment with ALF-Se NPs resulted in a significant upregulation of the pro-apoptotic gene p53 and downregulation of anti-apoptotic genes Bcl-2, MMP-9, and VEGF, indicating a shift toward pro-apoptotic signaling pathways.
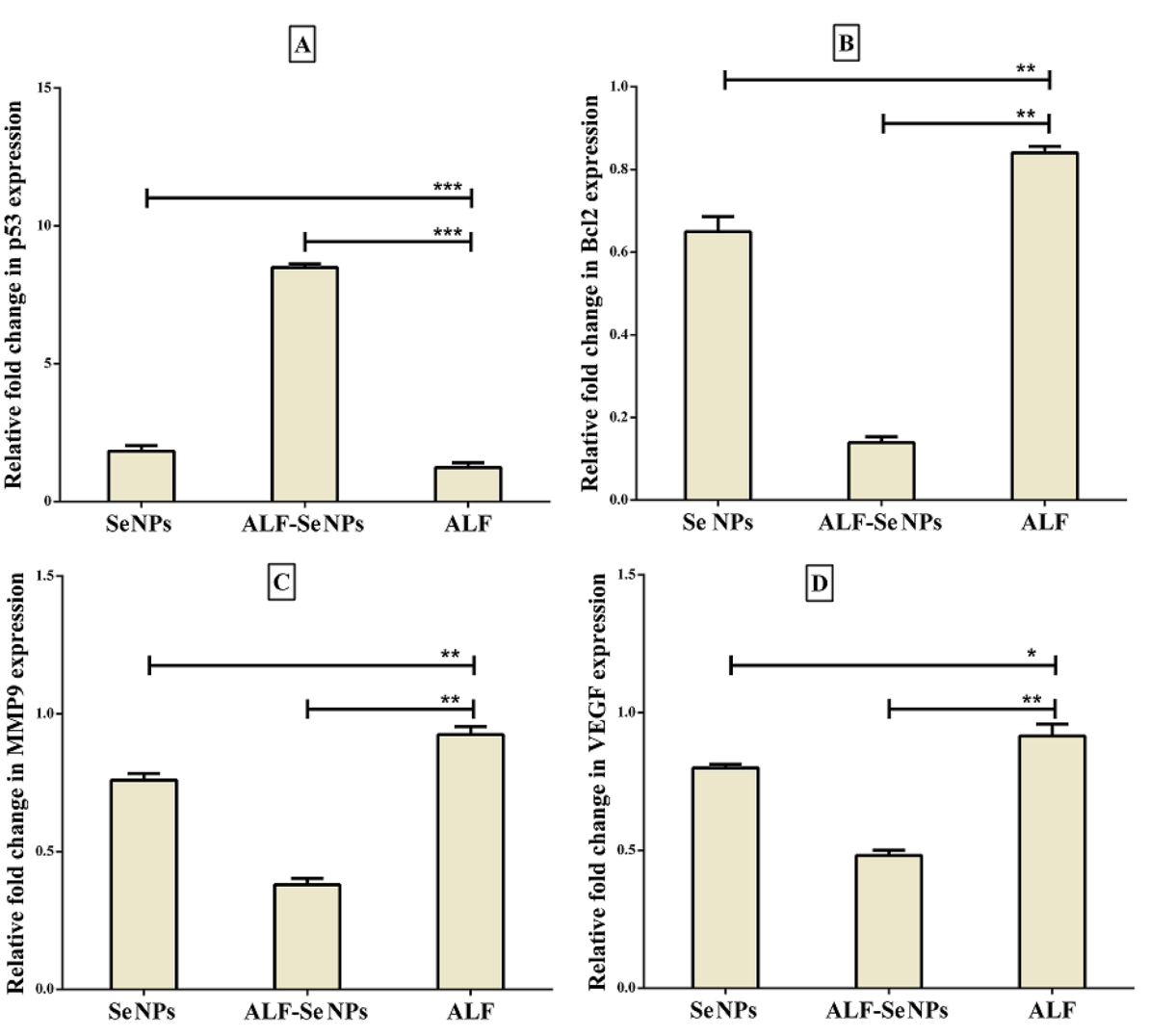
Figure 6. Relative fold change of gene expression in Caco2 cells afer treatment with ALF-Se NPs in comparison with ALF and Se NPs. (A) p53, (B) BCl2, © MMP9 and (D) VEGF. All values are demonstrated as mean ± SEM. ALF-Se NPs is statistically signifcant with ALF and Se NPs at p < 0.05*, p < 0.005** and p < 0.0005***. ALF; apolactoferrin, Se NPs; selenium nanoparticles, ALF-Se NPs; nanoformulation of apolactoferrin with Se NPs.
- Significance of the Result: This gene expression profile reinforces the concept that ALF-Se NPs can selectively induce apoptosis in cancer cells, supporting the development of targeted therapies that minimize adverse effects on normal cells.
- Key Innovations: The integration of gene expression analysis with the cytotoxicity studies provides a deeper insight into the biological mechanisms at play, establishing a clearer link between the observed cytotoxic effects and the underlying molecular changes.
Conclusion
The study concludes that lactoferrin-coated biosynthesized selenium nanoparticles exhibit promising anticancer activity, with a notable capacity to selectively target and induce apoptosis in multiple cancer cell types while sparing normal cells. The findings suggest that ALF-Se NPs could serve as a novel therapeutic agent with enhanced efficacy and reduced side effects compared to conventional therapies.
However, the study acknowledges certain limitations, including the need for further in vivo studies to validate these findings in a clinical setting. Future research should also focus on optimizing the formulation for improved stability and exploring the potential synergistic effects with other therapeutic agents to enhance anticancer efficacy further.
Reference:
El‑Fakharany, Esmail M., et al. “Anticancer activity of lactoferrin-coated biosynthesized selenium nanoparticles for combating different human cancer cells via mediating apoptotic effects.” Scientific Reports 13.1 (2023): 9579.
