Editor: Nina
Scientists develop a photocrosslinking spherical hydrogel-encapsulated targeting peptide-modified engineered exosome system to enhance drug delivery and therapeutic efficacy for osteoarthritis treatment.
Key Preview
Research Question
How can engineered exosomes encapsulated in hydrogels enhance drug delivery and therapeutic efficacy for osteoarthritis (OA)?
Research Design and Strategy
The study developed a novel photocrosslinking spherical gelatin methacryloyl hydrogel (GelMA) to encapsulate cartilage affinity peptide-modified engineered exosomes to improve drug delivery for OA therapy.
Method
The engineered exosomes were loaded with the small inhibitor LRRK2-IN-1 and tested for targeting capability, anti-inflammatory effects, and cartilage repair in vitro and in vivo.
Key Results
The engineered exosomes demonstrated enhanced targeting to chondrocytes, suppressed inflammation, and promoted cartilage repair in murine models, showing superior efficacy compared to conventional treatments.
Significance of the Research
This research provides a novel strategy for OA treatment, addressing limitations of existing therapies by improving drug retention and targeting within joint tissues.
Introduction
Osteoarthritis (OA) is a widespread degenerative joint disease characterized by the progressive deterioration of articular cartilage, leading to pain, inflammation, and functional impairment. It affects millions worldwide, particularly the elderly, and poses significant challenges to healthcare systems due to its prevalence and the associated burden of chronic pain and disability. The disease process involves a complex interplay of mechanical, biochemical, and genetic factors that contribute to cartilage degradation and joint inflammation.
Traditionally, the management of OA has relied on pharmacological interventions, including nonsteroidal anti-inflammatory drugs (NSAIDs) and corticosteroids, which are often administered systemically or through intra-articular injections. These treatments aim to alleviate pain and reduce inflammation, but they commonly fail to address the underlying causes of cartilage degeneration. Furthermore, conventional drug delivery methods often result in low drug retention at the site of action, leading to suboptimal therapeutic outcomes and necessitating frequent dosing.
One of the primary challenges associated with current treatment strategies is the rapid clearance of drugs from the joint, which limits their effectiveness. This rapid turnover results in insufficient drug concentrations within the affected cartilage, thereby failing to provide long-lasting relief or promote tissue repair. Additionally, the dense extracellular matrix of cartilage acts as a barrier to drug penetration, further complicating effective delivery to target cells.
In response to these challenges, innovative drug delivery strategies have emerged, focusing on enhancing the specificity and retention of therapeutic agents within the joint. One promising approach is the use of engineered exosomes—naturally occurring extracellular vesicles that can encapsulate drugs and target specific cells. By modifying the surface of these exosomes with cartilage-affinity peptides and encapsulating them in a hydrogel matrix, researchers aim to create a drug delivery system that improves retention, enhances targeting capability, and ultimately promotes cartilage repair in OA patients. This innovative strategy promises to overcome the limitations of traditional treatments, offering a more effective and sustained therapeutic solution for individuals suffering from osteoarthritis.
Research Team and Aim
The research team was led by Junlai Wan, along with co-authors Zhiyi He, Renpeng Peng, and others from Huazhong University of Science and Technology. This research was conducted in 2023 and culminated in the publication of their findings in the paper titled “Injectable photocrosslinking spherical hydrogel-encapsulated targeting peptide-modified engineered exosomes for osteoarthritis therapy” in the Journal of Nanobiotechnology.
The aim of the research, as articulated by the lead researcher, Junlai Wan, was to investigate the potential of a novel exosome-based hydrogel system to improve drug delivery and therapeutic outcomes in osteoarthritis. Specifically, the study sought to address the limitations of existing therapies by enhancing drug retention and targeting within joint tissues, ultimately contributing to more effective treatment strategies for OA patients.
Experimental Process
Primary Technique: Exosome Isolation and Modification
The primary technique utilized in this study was the isolation and modification of exosomes derived from rat bone marrow mesenchymal stromal cells (BMSCs). This foundational step was essential for creating engineered exosomes that could effectively deliver therapeutic agents to target cartilage in osteoarthritis (OA) treatment.
Experiment 1: Isolation and Surface Modification of Exosomes from Rat Bone Marrow Mesenchymal Stromal Cells
Key Steps:
- Cell Culture: BMSCs were cultured in DMEM medium supplemented with 10% fetal bovine serum (FBS) until they reached 30-50% confluency.
- Exosome Isolation: The culture medium was replaced with DMEM without FBS and incubated for 48–72 hours. The supernatant was collected and subjected to a series of centrifugation steps: first at 300 g for 10 minutes to remove cells, then at 2000 g for 10 minutes to eliminate dead cells, followed by 10,000 g for 30 minutes to remove cell debris. Finally, the supernatant underwent ultracentrifugation at 100,000 g for 70 minutes to isolate the exosomes.
- Characterization: The exosomes were characterized for purity and concentration using Western blot analysis of specific exosomal markers (TSG101, CD81, CD9) and nanoparticle tracking analysis for size distribution.
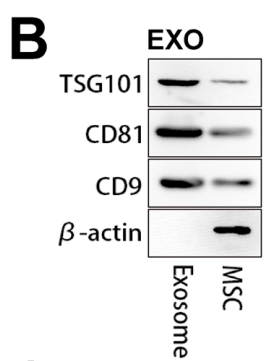
Figure 1. Western blot analyses showing that MSC exosomes could express characteristic marker proteins, such as TSG101, CD81, and CD9.
Data Collection and Analysis:
Data were collected by assessing the protein concentration of isolated exosomes using a BCA assay. The results were analyzed to ensure the successful isolation of high-purity exosomes suitable for subsequent modifications and applications.
Novel Aspects:
This study introduced a novel surface modification technique using methacrylic anhydride to enhance the targeting capabilities of exosomes. By amidating the surface of exosomes, a coupling with cartilage affinity peptides (WYRGRL) was achieved, which significantly improved their targeting efficiency to chondrocytes compared to traditional methods that did not utilize such targeted modifications.
Experiment 2: Hydrogel Formation and Exosome Encapsulation
The second phase involved the creation of a photocrosslinking hydrogel to encapsulate the modified exosomes, thereby enhancing their retention and delivery capabilities within the joint.
Key Steps:
- Hydrogel Preparation: A 20% solution of gelatin methacryloyl (GelMA) was prepared by dissolving the polymer in phosphate-buffered saline (PBS) at 40 °C.
- Exosome Mixing: The peptide-modified exosomes were added to the GelMA solution at a concentration of 300 µg/mL and mixed thoroughly.
- Photocrosslinking: The mixture was then emulsified with a liquid paraffin span-80 mixture in a round-bottom flask, followed by the addition of a photoinitiator (200 µL LAP) and exposure to ultraviolet light for crosslinking.
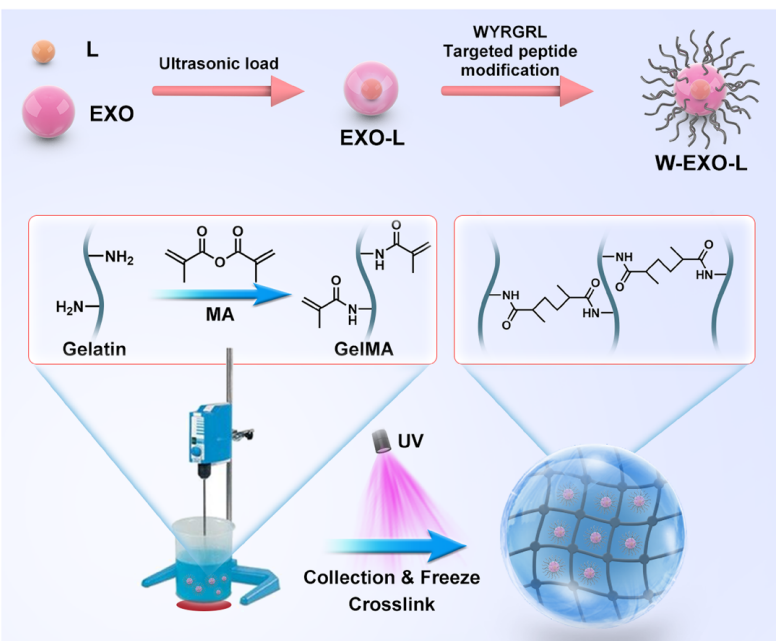
Figure 2. The protocol of W-Exo-L@GelMA preparation. L, LRRK2-IN-1; EXO, exosome; W, cartilage afnity WYRGRL (W) peptide; UV, ultraviolet
Data Collection and Analysis:
The encapsulation efficiency of exosomes within the GelMA hydrogel was determined using a protein quantification assay after disrupting the hydrogel matrix. A release profile of the therapeutic agent (LRRK2-IN-1) from the hydrogel was generated using high-performance liquid chromatography (HPLC) to assess the release kinetics over time.
Novel Aspects:
The integration of photocrosslinking with exosome encapsulation represents a significant advancement over traditional drug delivery systems, which often lack the structural integrity and controlled release properties necessary for sustained therapeutic efficacy. This hydrogel system allows for a prolonged release of the exosome-encapsulated drug, enhancing its therapeutic potential.
Experiment 3: In Vitro Evaluation of Therapeutic Efficacy
The third experiment focused on evaluating the therapeutic efficacy of the engineered exosome-loaded hydrogels in vitro, particularly their effects on inflammatory and catabolic responses in chondrocytes.
Key Steps:
- Chondrocyte Isolation: Chondrocytes were isolated from the knee joints of C57BL/6 mice and cultured in DMEM with 10% FBS until reaching 80% confluency.
- IL-1β Treatment: The chondrocytes were treated with IL-1β (5 ng/mL) to induce inflammatory conditions.
- Exosome Treatment: Subsequently, the chondrocytes were treated with varying concentrations of W-Exo-L@GelMA loaded with LRRK2-IN-1 for 48 hours.
- Western Blot Analysis: After treatment, the cells were harvested, and protein extracts were analyzed via Western blotting to quantify the expression levels of anabolic (COL2, SOX9) and catabolic (MMP3, MMP13, COX2, iNOS) markers.
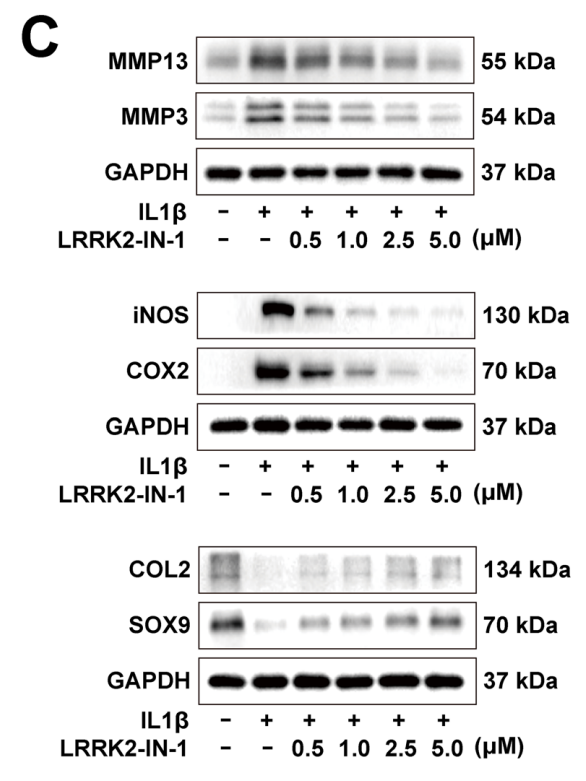
Figure 3. Western blot analyses of the protein levels of anabolic, catabolic, infammatory factors in the IL-1β-induced chondrocytes treated with 0.5, 1.0, 2.5, and 5.0 µM LRRK2-IN-1 for 24 h. LRRK2-IN-1 suppressed MMP3, MMP13, iNOS, and COX2 and induced COL2 and SOX9 in a dose-dependent manner.
Data Collection and Analysis:
Data were collected through densitometric analysis of the Western blot bands using ImageJ software. Statistical comparisons between treated and untreated groups were performed using one-way ANOVA.
Novel Aspects:
This experiment demonstrated the superior anti-inflammatory and anabolic effects of the engineered exosomes compared to traditional treatments. The use of exosomes for drug delivery not only provides the therapeutic agent but also utilizes the inherent properties of exosomes to mitigate inflammation and promote cartilage repair, marking a significant improvement over conventional drug delivery systems.
Experiment 4: In Vivo Efficacy in Osteoarthritis Models
The final experiment assessed the efficacy of W-Exo-L@GelMA in a murine model of osteoarthritis, evaluating its therapeutic potential and joint retention capabilities.
Key Steps:
- Surgical Induction of OA: Osteoarthritis was induced in male C57BL/6 mice (8 weeks old) via destabilization of the medial meniscus (DMM) procedure.
- Intra-Articular Injection: Mice were randomly assigned to receive intra-articular injections of either saline, L@GelMA, Exo@GelMA, Exo-L@GelMA, or W-Exo-L@GelMA (10 µL of each treatment containing 800 µg/mL LRRK2-IN-1) every two weeks for a total of eight weeks.
- Post-treatment Analysis: After the treatment period, knee joints were harvested for micro-computed tomography (µCT) and histological evaluations using safranin O/fast green staining to assess cartilage integrity and bone morphology.
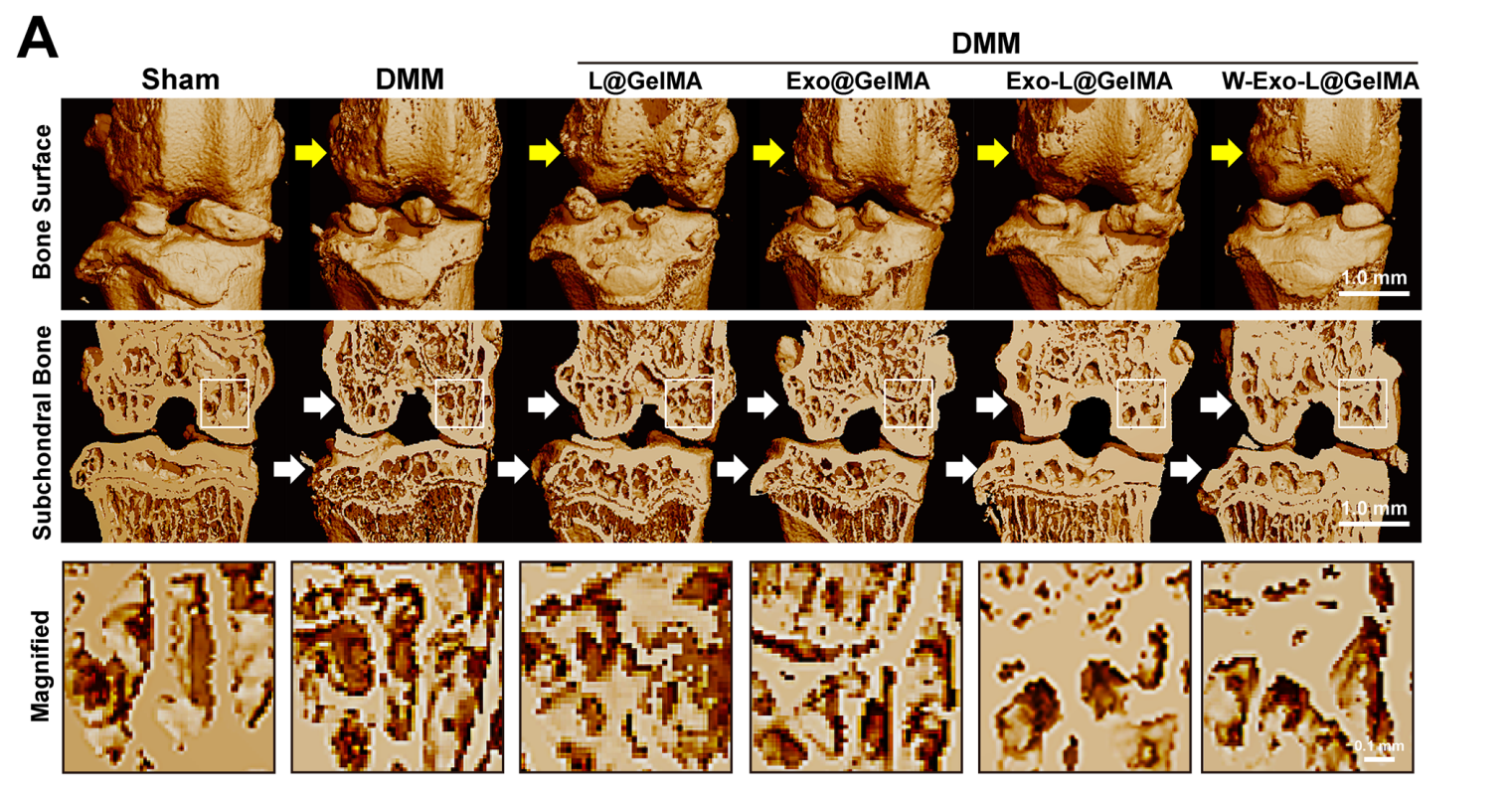
Figure 4. Micro-CT representative images of the bone surface and the coronal plane of the subchondral bone of joints. W-Exo-L@GelMA notably reduced the osteophyte formation of the surface and the loss of subchondral bone.
Data Collection and Analysis:
Data were collected through µCT imaging to visualize bone structure and histological scoring to quantify cartilage damage. Statistical analyses were performed to compare the therapeutic effects across treatment groups.
Novel Aspects:
The in vivo study demonstrated the enhanced retention and therapeutic efficacy of the W-Exo-L@GelMA system in a clinically relevant model of OA. This approach to targeted delivery and retention of therapeutic agents represents a significant advancement over traditional methods, offering prolonged therapeutic effects and improved outcomes in cartilage repair.
Conclusion
The successful development of the drug delivery system, W-Exo-L@GelMA, was achieved through a combination of innovative strategies that enhanced the targeting ability and retention of engineered exosomes. By modifying the surface of exosomes with a cartilage-affinity peptide and encapsulating them within a photocrosslinking hydrogel, the researchers effectively overcame the limitations associated with rapid drug clearance and low retention rates typically seen in conventional treatments for osteoarthritis.
The highlights of the study include the demonstration that the engineered exosome-loaded hydrogel significantly improved drug delivery to chondrocytes, suppressed inflammation, and promoted cartilage repair in both in vitro and in vivo models. Additionally, the findings indicate that this novel approach not only enhances therapeutic efficacy but also offers a promising avenue for more effective, personalized treatments for individuals suffering from osteoarthritis. The research underscores the potential of integrating nanotechnology with biological engineering to create advanced therapeutic solutions for complex diseases.
Reference:
Wan, Junlai, et al. “Injectable photocrosslinking spherical hydrogel-encapsulated targeting peptide-modified engineered exosomes for osteoarthritis therapy.” Journal of Nanobiotechnology, vol. 21, no. 284, 2023.
