Editor: Nina
Scientists synthesize amphiphilic polypeptides through post-polymerization modification to create effective nanoparticles for the targeted delivery of hydrophobic drugs, such as paclitaxel, enhancing their solubility and cytostatic efficacy against lung cancer cells.
Key Preview
- Research Question: The study investigates the synthesis and evaluation of amphiphilic polypeptides derived from poly(α,L-glutamic acid) as delivery systems for hydrophobic drugs, particularly focusing on their efficacy in encapsulating paclitaxel (PTX).
- Research Design and Strategy: The research employs a post-polymerization modification approach to create amphiphilic copolymers, followed by characterizing their ability to form nanoparticles suitable for drug delivery.
- Method: The researchers synthesized poly(α,L-glutamic acid) and modified it with various hydrophobic amino acids and D-glucosamine. The resulting nanoparticles were characterized for size, surface charge, cytotoxicity, and drug loading capacity.
- Key Results: The synthesized nanoparticles demonstrated a size range of 170-330 nm, showed low toxicity to human lung epithelial cells, and exhibited significant cytostatic efficacy against human lung adenocarcinoma cells (A549).
- Significance of the Research: This study highlights the potential of biocompatible and biodegradable amphiphilic polypeptides as effective delivery systems for hydrophobic drugs, addressing challenges in drug solubility and bioavailability.
Introduction
Lung cancer remains one of the leading causes of cancer-related deaths worldwide, characterized by its aggressive nature and high mortality rates. The disease is primarily classified into two major types: non-small cell lung cancer (NSCLC) and small cell lung cancer (SCLC). Among these, NSCLC accounts for approximately 85% of all cases, and its treatment often involves a combination of surgery, radiation therapy, and chemotherapy. Despite advances in medical technology, the prognosis for lung cancer patients remains poor, with many patients experiencing recurrence and metastasis.
Traditionally, the delivery of chemotherapeutic agents, such as paclitaxel (PTX), in lung cancer treatment has primarily relied on systemic administration methods. These approaches typically involve intravenous infusion or oral ingestion, allowing the drug to circulate throughout the body to target cancer cells. However, this method presents significant limitations, including low solubility of hydrophobic drugs, non-specific distribution, and severe side effects due to toxicity to healthy tissues. As a result, patients often experience reduced therapeutic efficacy and increased adverse reactions, leading to challenges in achieving optimal treatment outcomes.
The current strategies in drug delivery for lung cancer treatment face several distinct challenges. One major issue is the poor bioavailability of hydrophobic drugs like PTX, which limits their therapeutic potential. Furthermore, the rapid clearance of drugs from the bloodstream and their uptake by non-target cells, such as macrophages, can lead to suboptimal dosing and reduced effectiveness against tumors. These challenges contribute to the development of drug resistance and can significantly hinder the overall success of cancer therapies.
To address these issues, innovative drug delivery strategies have emerged, focusing on the design of advanced nanocarriers that enhance the solubility, stability, and targeted delivery of chemotherapeutic agents. One promising approach involves the use of amphiphilic polypeptides that can encapsulate hydrophobic drugs and form nanoparticles. These nanoparticles not only improve drug solubility and bioavailability but also offer the potential for passive and active targeting of cancer cells, thereby minimizing systemic toxicity and enhancing therapeutic efficacy. By leveraging the properties of biodegradable and biocompatible materials, this innovative drug delivery strategy aims to overcome the limitations of traditional methods, paving the way for more effective treatments in lung cancer and other malignancies.
Research Team and Aim
The research was conducted by a collaborative team led by Evgenia G. Korzhikova-Vlakh, a prominent researcher affiliated with the Institute of Macromolecular Compounds at the Russian Academy of Sciences. The study took place over the course of 2022 and culminated in the publication of the paper titled “Amphiphilic Polypeptides Obtained by the Post-Polymerization Modification of Poly(Glutamic Acid) and Their Evaluation as Delivery Systems for Hydrophobic Drugs.” This research was published in the International Journal of Molecular Sciences in January 2023.
The aim of the research, as articulated by Korzhikova-Vlakh, was to synthesize novel amphiphilic polypeptides through post-polymerization modification and evaluate their properties as effective drug delivery systems for hydrophobic drugs, particularly focusing on improving the solubility and bioavailability of paclitaxel.
Experimental Process
Primary Technique
The primary technique employed in this study was the post-polymerization modification of poly(α,L-glutamic acid) (P[E]) to create amphiphilic copolymers, which were then evaluated as delivery systems for hydrophobic drugs such as paclitaxel (PTX). This approach allows for the tailoring of polypeptide properties to enhance drug encapsulation efficiency and biocompatibility.
Synthesis of Poly(α,L-Glutamic Acid)
- Preparation of Monomer: The N-carboxyanhydride (NCA) of L-glutamic acid γ-benzyl ester was synthesized using triphosgene and L-glutamic acid.
- Ring-Opening Polymerization (ROP): A solution of the synthesized NCA (4 wt%) in anhydrous 1,4-dioxane was polymerized at 30 °C for 72 hours using n-hexylamine as an initiator. The monomer-to-initiator ratio was maintained at 100:1.
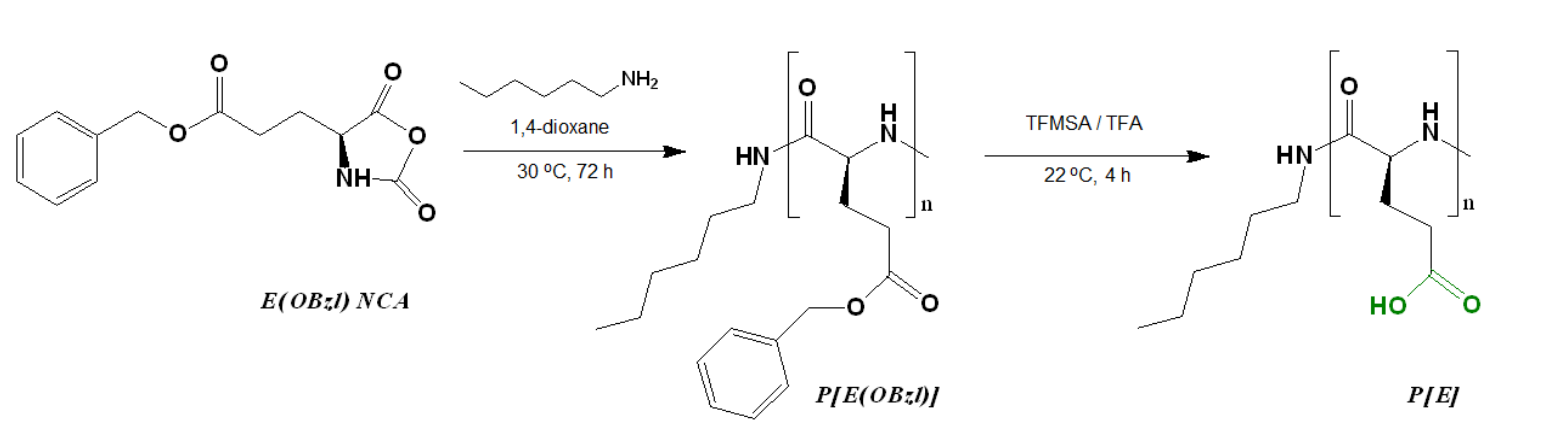
Figure 1. Scheme of the synthesis of poly(α‐glutamic acid) by the ring‐opening polymerization of the N‐carboxyanhydride of L‐glutamic acid γ‐benzyl ester.
- Deprotection: The resulting copolymer was subjected to acidic hydrolysis using trifluoroacetic acid and trifluoromethanesulfonic acid to remove benzyl protective groups, confirming a 94% removal via NMR spectroscopy.
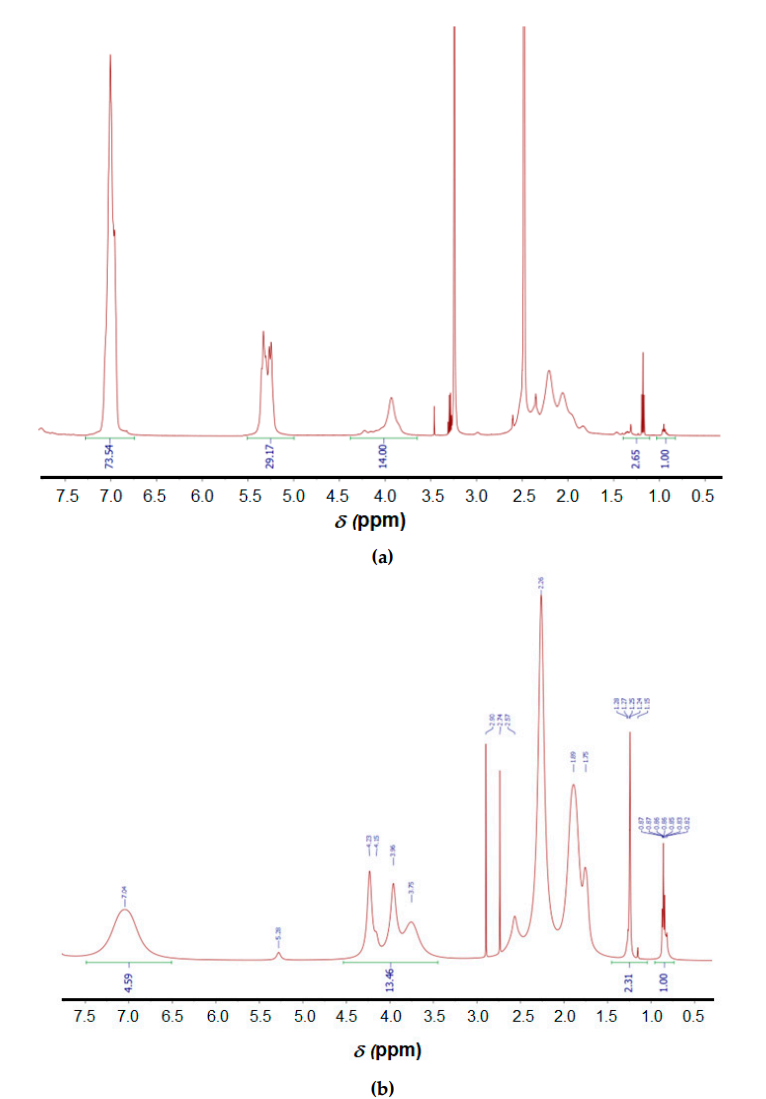
Figure 2. 1H NMR spectra of Bzl-protected (a) and deprotected (b) poly(α,L-glutamic acid) (DMSO-d6, 25 °C): Signals (ppm): 0.80-0.85 (СН3, hexylamine), 1.00-1.33 (СН2, hexylamine), 3.5-4.5 (СН, Glu), 5.0-5.5 (O-CH2-C6H5, Glu(OBzl)), 6.7-7.4 (O-CH2-C6H5, Glu(OBzl)).
Post-Polymerization Modification
- Activation of Carboxyl Groups: The deprotected P[E] was activated using N,N′-diisopropylcarbodiimide (DIC) and N-hydroxysuccinimide (NHS) to form activated esters. This step was crucial for enabling the subsequent reaction with amino acids.
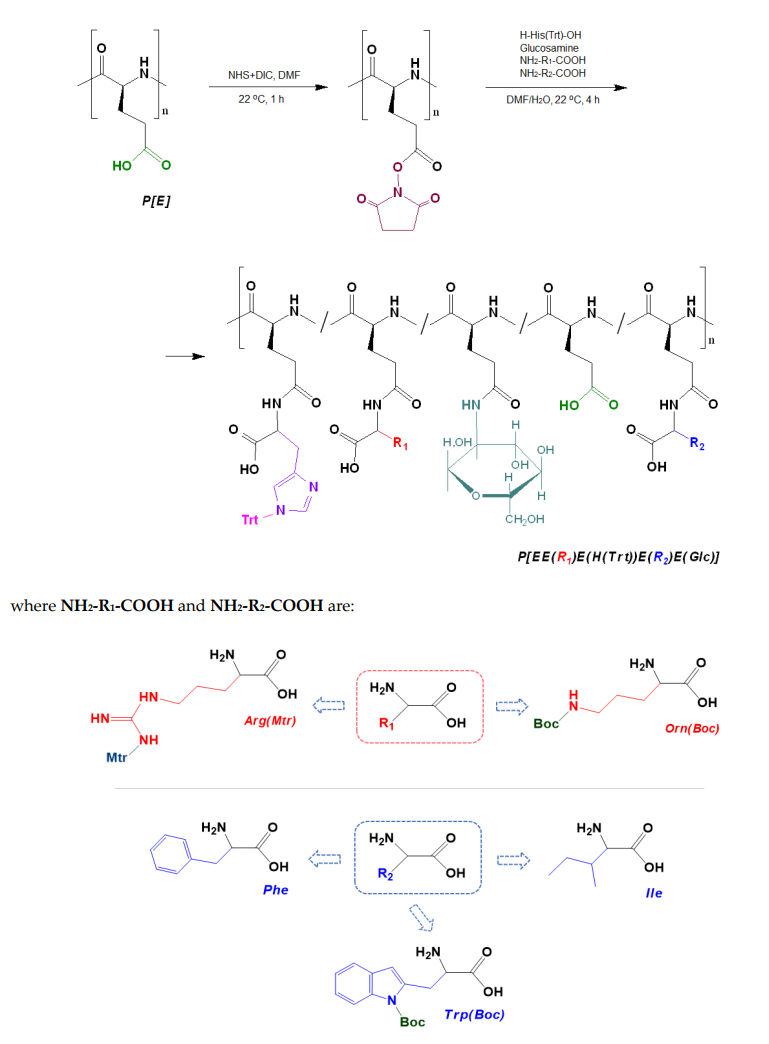
Figure3.Scheme of the synthesis of amphiphilic copolymers by the post‐polymerization modifica‐ tion of poly(α‐glutamic acid) and structures of amino acid derivatives used for modification: R1 (red moiety) indicates a basic amino acid, R2 (blue moiety) indicates a hydrophobic amino acid.
- Modification with Amino Acids and D-Glucosamine: A four-component mixture of amino acids (L-phenylalanine, L-isoleucine, L-tryptophan, L-ornithine, and L-arginine) and D-glucosamine (in specified molar ratios) was added to the activated P[E] solution. The reaction was stirred for 4 hours at room temperature in a DMF/water mixture (70/30 v/v) for hydrophobic amino acids and 80/20 for others.
- Purification: The modified copolymers were purified using dialysis against DMF and deionized water to remove unreacted amino acids, followed by freeze-drying.
Characterization of Nanoparticles
- Nanoparticle Formation: Depending on the hydrophobic amino acid composition, nanoparticles were formed either via self-assembly during a slow gradient phase inversion (dialysis from DMF to water) or by nanoprecipitation for tryptophan-containing polymers.
- Physicochemical Characterization: Dynamic light scattering (DLS) and nanoparticle tracking analysis (NTA) were employed to measure the hydrodynamic diameter (DH), polydispersity index (PDI), and ζ-potential of the nanoparticles to assess their size distribution and surface charge.
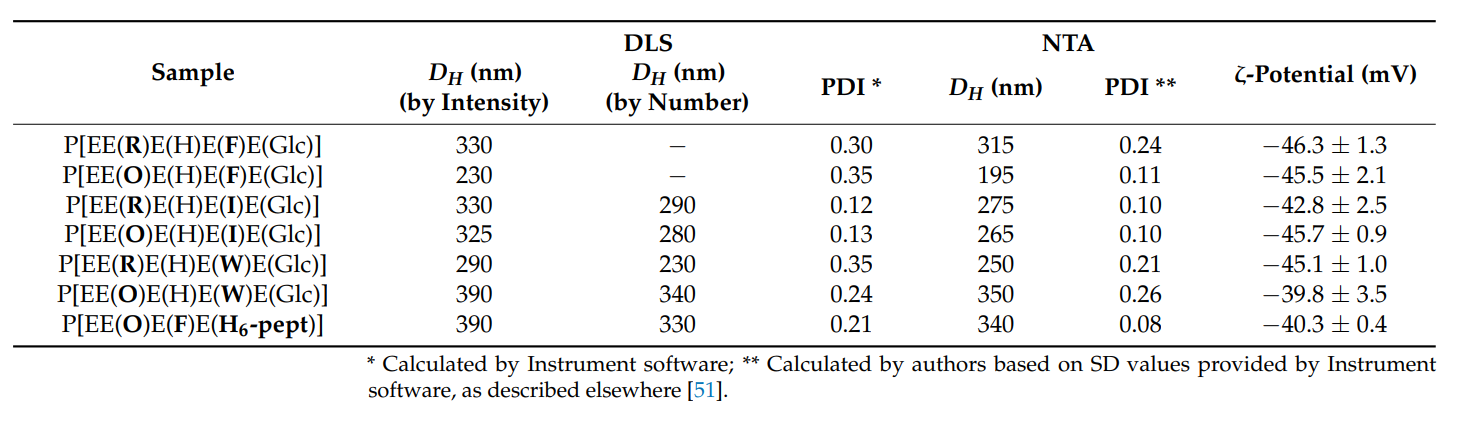
Table 1. Characteristics of nanoparticles obtained from the synthesized amphiphilic polypeptides
Drug Loading and Evaluation
- Paclitaxel Loading: PTX was incorporated into the nanoparticles by dissolving a predetermined amount of PTX (50-200 µg/mg of polymer) in DMSO, followed by freeze-drying and subsequent dispersion in buffer.
- Characterization of Drug Encapsulation: The efficiency of drug loading was determined by quantifying free PTX using HPLC after purification via ultrafiltration, and the hydrodynamic diameter of the loaded nanoparticles was measured again to assess any changes post-loading.
Data Collection and Analysis
Data was collected through various assays, including DLS for size measurements and HPLC for quantifying drug loading. Statistical analyses were performed using one-way ANOVA to evaluate differences between groups, ensuring that results were rigorously analyzed for significance.
Novel Aspects and Advantages
This research introduced a novel post-polymerization modification approach that provides enhanced control over the functionalization of polypeptides compared to traditional methods like ring-opening polymerization. The ability to incorporate hydrophobic amino acids and glucosamine into the polymer backbone allows for the formation of nanoparticles with improved drug loading capacity, biocompatibility, and reduced macrophage uptake, addressing common challenges associated with conventional delivery systems. The synthesized nanoparticles demonstrated a high encapsulation efficiency and cytotoxicity against cancer cells while maintaining low toxicity to normal lung epithelial cells, showcasing their potential for therapeutic applications in drug delivery.
This approach not only enhances the flexibility in designing drug delivery systems but also improves overall drug bioavailability, making it a significant advance in the field of polymer-based nanomedicine. for cytotoxicity against human lung epithelial cells (BEAS-2B), confirming their non-toxic nature. Additionally, the research demonstrated effective loading of paclitaxel, with encapsulation efficiencies reaching up to 99%.
Conclusion
The successful development of this drug delivery system was achieved through the innovative synthesis of amphiphilic polypeptides via post-polymerization modification of poly(α,L-glutamic acid). This method allowed for the effective incorporation of hydrophobic amino acids and D-glucosamine, which facilitated the formation of nanoparticles capable of encapsulating hydrophobic drugs like paclitaxel.
The study highlighted several key findings, including the non-toxic nature of the synthesized nanoparticles to human lung epithelial cells (BEAS-2B), their significant cytostatic efficacy against human lung adenocarcinoma cells (A549), and their ability to achieve high encapsulation efficiency for paclitaxel. Overall, this research demonstrates the potential of the developed amphiphilic polypeptide-based nanoparticles as effective delivery systems that can significantly improve the solubility and bioavailability of hydrophobic drugs, addressing critical challenges in cancer treatment.
Reference:
Dzhuzha, Apollinariia Yu, et al. “Amphiphilic polypeptides obtained by the post-polymerization modification of poly (glutamic acid) and their evaluation as delivery systems for hydrophobic drugs.” International Journal of Molecular Sciences 24.2 (2023): 1049.
